Search
Thermo Scientific Chemicals
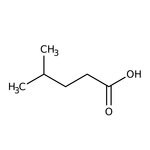
Ácido 4-metilvalérico, 99 %, Thermo Scientific Chemicals
CAS: 646-07-1 | C6H12O2 | 116.16 g/mol
Número de catálogo A15405.22
también denominado A15405-22
Precio (EUR)
154,00
Each
Cantidad:
100 g
Precio (EUR)
154,00
Each
Identificadores químicos
CAS646-07-1
IUPAC Name4-methylpentanoic acid
Molecular FormulaC6H12O2
InChI KeyFGKJLKRYENPLQH-UHFFFAOYSA-N
SMILESCC(C)CCC(O)=O
Ver más
Especificaciones Hoja de especificaciones
Hoja de especificaciones
Refractive Index1.4125-1.4155 @ 20?C
Assay (Aqueous acid-base Titration)≥98.5 to ≤101.5%
FormLiquid
Identification (FTIR)Conforms
Appearance (Color)Clear colorless
Ver más
4-Methylvaleric acid has been used in asymmetric synthesis of (4S)-4-isopropyl-3-[(2?S)-2?,4?-dimethyl-valeryl)]-2-oxazolidinone. It is a versatile building block used in the synthesis of various pharmaceutical and biologically active compounds, including inhibitors and antagonists. It is involved in the synthesis of novel series of high affinity, functionally potent antagonists of the CCR1 receptor.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Aplicaciones
el ácido 4-metilvalérico se ha utilizado en la síntesis asimétrica de (4S)-4-isopropil-3-[(2′S)-2′,4′-dimetil-valeril)]-2-oxazolidinona. Es un elemento básico versátil utilizado en la síntesis de varios compuestos farmacéuticos y biológicamente activos, incluyendo inhibidores y antagonistas. Interviene en la síntesis de nuevas series de antagonistas funcionalmente potentes y de alta afinidad del1 receptor CCR.
Solubilidad
soluble en alcohol. Ligeramente soluble en agua.
Notas
almacénese en condiciones frescas y secas en un recipiente hermético. Almacénese lejos de la humedad, los agentes oxidantes y las bases.
el ácido 4-metilvalérico se ha utilizado en la síntesis asimétrica de (4S)-4-isopropil-3-[(2′S)-2′,4′-dimetil-valeril)]-2-oxazolidinona. Es un elemento básico versátil utilizado en la síntesis de varios compuestos farmacéuticos y biológicamente activos, incluyendo inhibidores y antagonistas. Interviene en la síntesis de nuevas series de antagonistas funcionalmente potentes y de alta afinidad del1 receptor CCR.
Solubilidad
soluble en alcohol. Ligeramente soluble en agua.
Notas
almacénese en condiciones frescas y secas en un recipiente hermético. Almacénese lejos de la humedad, los agentes oxidantes y las bases.
RUO – Research Use Only
General References:
- Kazutoku Ohta; Atsuya Towata; Masayoshi Ohashi; Toyohide Takeuchi. Separation and conductimetric detection of C1-C7 aliphatic monocarboxylic acids and C1-C7 aliphatic monoamines on unfunctionized polymethacrylate resin columns. Journal of Chromatography A. 2004, 1039, (1-2), 171-177.
- Vincent Guerlavais; Patrick J. Carroll; Madeleine M. Joullié. Progress towards the total synthesis of callipeltin A. Asymmetric synthesis of (2R, 3R, 4S)-3-hydroxy-2, 4, 6-trimethylheptanoic acid. Tetrahedron: Asymmetry. 2002, 13(7), 675-680.