Search
Thermo Scientific Chemicals
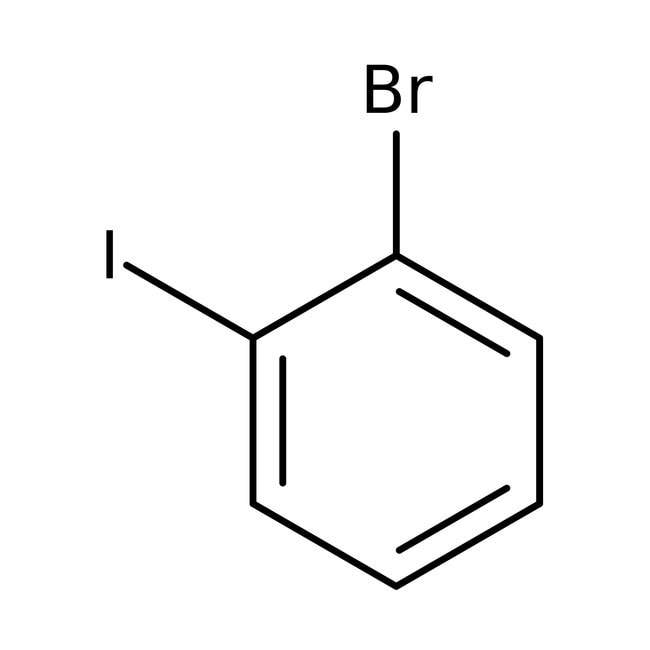
1-Bromo-2-yodobeceno, +98 %, +estab. con cobre, +Thermo Scientific Chemicals
CAS: 583-55-1 | C6H4BrI | 282.906 g/mol
Número de catálogo A17020.06
también denominado A17020-06
Precio (EUR)
33,70
Each
Cantidad:
5 g
Precio (EUR)
33,70
Each
Identificadores químicos
CAS583-55-1
IUPAC Name1-bromo-2-iodobenzene
Molecular FormulaC6H4BrI
InChI KeyOIRHKGBNGGSCGS-UHFFFAOYSA-N
SMILESBrC1=CC=CC=C1I
Ver más
Especificaciones Hoja de especificaciones
Hoja de especificaciones
Refractive Index1.6625-1.6675 @ 20?C
Appearance (Color)Clear colorless to yellow to brown
Identification (FTIR)Conforms
Assay (GC)≥98.0%
FormLiquid
2-Bromoiodobenzene is used in the synthesis of diarylamines.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Aplicaciones
2-el bromoyodobenceno se utiliza en la síntesis de diarilaminas.
Solubilidad
no miscible o difícil de mezclar en agua.
Notas
sensible a la luz. Proteja el producto de la luz. Almacénese lejos de los agentes oxidantes.
2-el bromoyodobenceno se utiliza en la síntesis de diarilaminas.
Solubilidad
no miscible o difícil de mezclar en agua.
Notas
sensible a la luz. Proteja el producto de la luz. Almacénese lejos de los agentes oxidantes.
RUO – Research Use Only
General References:
- María E Budén.; Victoria A Vaillard.; Sandra E Martin.; Roberto A Rossi. Synthesis of carbazoles by intramolecular arylation of diarylamide anions.J Org Chem.2009,74(12), 4490-4498.
- Biao Lu.; Bao Wang.; Yihua Zhang.; and Dawei Ma. CuI-Catalyzed Domino Process to 2,3-Disubstituted Benzofurans from 1-Bromo-2-Iodobenzenes and β-Keto Esters.J. Org. Chem.,2007,72,(14), 5337-5341.
- Benzyne precursor. In the presence of excess of a Grignard reagent, the initially formed benzyne undergoes nucleophilic addition to give an ortho-substituted phenylmagnesium halide: Synth. Commun., 18, 1749 (1988).
- Regioselective Suzuki coupling with arylboronic acids has been achieved giving 2-bromobiphenyl derivatives: Synlett, 207 (1992).