Search
Thermo Scientific Chemicals
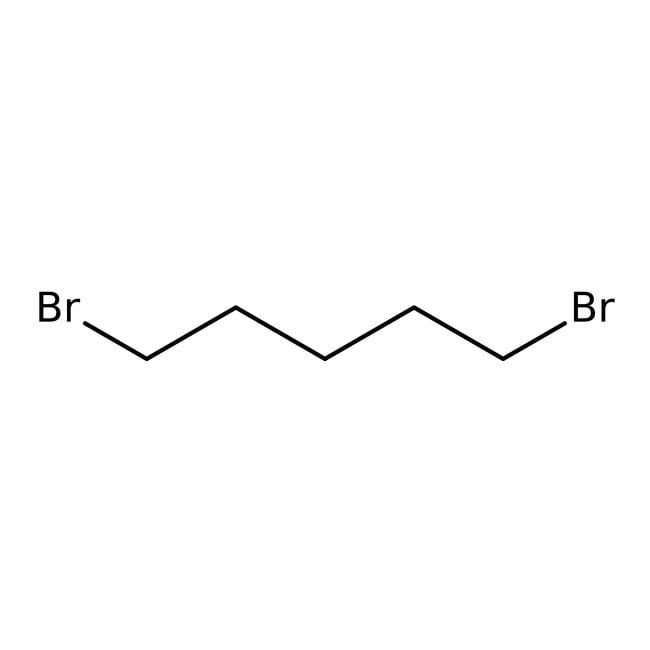
1,5-Dibromopentano, 98 %, Thermo Scientific Chemicals
CAS: 111-24-0 | C5H10Br2 | 229.943 g/mol
Número de catálogo A18465.22
también denominado A18465-22
Precio (EUR)
33,60
Each
Cantidad:
100 g
Precio (EUR)
33,60
Each
Identificadores químicos
CAS111-24-0
IUPAC Name1,5-dibromopentane
Molecular FormulaC5H10Br2
InChI KeyIBODDUNKEPPBKW-UHFFFAOYSA-N
SMILESBrCCCCCBr
Ver más
Especificaciones Hoja de especificaciones
Hoja de especificaciones
Identification (FTIR)Conforms
Appearance (Color)Clear colorless to pale yellow
Refractive Index1.5090-1.5145 @ 20?C
Assay (GC)≥97.5%
FormLiquid
1,5-Dibromopentane is used in the preparation of 1,5-di-Grignard reagent and novel spirocyclic pyrrolidones. It acts as a growth supplement for Yarrowia lipolytica 3589, a tropical marine yeast. It is involved in the synthesis of N-alkylated piperidine as well as tetrydrothiopyran (thiane) by reaction with primary amine and sodium sulfide respectively.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Aplicaciones
El 1,5-dibromopentano se utiliza en la preparación de pirrolidonas espirocíclicas nuevas y 1,5-di-reactivos de Grignard. Actúa como un suplemento de crecimiento para Yarrowia lipolytica 3589, un hongo marino tropical. Participa en la síntesis de la piperidina N-alquilada y el tetridotiopirano (tiano) por reacción con amina primaria y sulfuro de sodio respectivamente.
Solubilidad
Miscible con agua, ligeramente miscible con cloroformo y benceno.
Notas
Incompatible con agentes oxidantes fuertes y bases fuertes.
El 1,5-dibromopentano se utiliza en la preparación de pirrolidonas espirocíclicas nuevas y 1,5-di-reactivos de Grignard. Actúa como un suplemento de crecimiento para Yarrowia lipolytica 3589, un hongo marino tropical. Participa en la síntesis de la piperidina N-alquilada y el tetridotiopirano (tiano) por reacción con amina primaria y sulfuro de sodio respectivamente.
Solubilidad
Miscible con agua, ligeramente miscible con cloroformo y benceno.
Notas
Incompatible con agentes oxidantes fuertes y bases fuertes.
RUO – Research Use Only
General References:
- Lifa, T.; Tieu, W.; Hocking, R. K.; Codd, R. Forward and Reverse (Retro) Iron(III) or Gallium(III) Desferrioxamine E and Ring-Expanded Analogues Prepared Using Metal-Templated Synthesis from endo-Hydroxamic Acid Monomers. Inorg. Chem. 2015, 54 (7), 3573-3583.
- Perroni, D. V.; Baez-Cotto, C. M.; Sorenson, G. P.; Mahanthappa, M. K. Linker Length-Dependent Control of Gemini Surfactant Aqueous Lyotropic Gyroid Phase Stability. J. Phys. Chem. Lett. 2015, 6 (6), 993-998.