Search
Thermo Scientific Chemicals
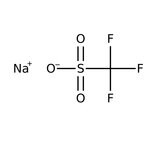
Trifluorometanosulfonato de sodio, 98 %, Thermo Scientific Chemicals
CAS: 2926-30-9 | CF3NaO3S | 172.053 g/mol
Número de catálogo B20776.06
también denominado B20776-06
Precio (EUR)
55,00
Each
Cantidad:
5 g
Precio (EUR)
55,00
Each
Identificadores químicos
CAS2926-30-9
IUPAC Namesodium trifluoromethanesulfonate
Molecular FormulaCF3NaO3S
InChI KeyXGPOMXSYOKFBHS-UHFFFAOYSA-M
SMILES[Na+].[O-]S(=O)(=O)C(F)(F)F
Ver más
Especificaciones Hoja de especificaciones
Hoja de especificaciones
Elemental Analysis(Fluorine, F) : 32.30-34.11%
FormPowder
Appearance (Color)White
Water Content (Karl Fischer Titration)<1.0%
Assay (unspecified)(based on F Elemental analysis) : >97.5 to <102.5&
Sodium trifluoromethanesulfonate is used in the preparation of N-fluoro-2-methylpyridinium triflate by reaction with dinitrogen difluoride as a reagent. It is also used as a chaotropic mobile phase additive in reversed-phase liquid chromatography (RP-LC).
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Aplicaciones
El trifluorometanosulfonato de sodio se utiliza en la preparación del triflato de N-fluoro-2-metilpiridinio por reacción con difluoruro de dinitrógeno como reactivo. También se utiliza como aditivo de fase móvil caotrópico en cromatografía líquida de fase reversa (RP-LC).
Solubilidad
Soluble en agua.
Notas
Higroscópico. Incompatible con ácidos, bases y agentes oxidantes fuertes.
El trifluorometanosulfonato de sodio se utiliza en la preparación del triflato de N-fluoro-2-metilpiridinio por reacción con difluoruro de dinitrógeno como reactivo. También se utiliza como aditivo de fase móvil caotrópico en cromatografía líquida de fase reversa (RP-LC).
Solubilidad
Soluble en agua.
Notas
Higroscópico. Incompatible con ácidos, bases y agentes oxidantes fuertes.
RUO – Research Use Only
General References:
- Sulfinylation of aromatic compounds can be brought about in triflic acid at room temperature to give good yields of aryl trifluoromethyl sulfoxides: Synlett, 550 (2001).
- Catalyzes the aza Diels-Alder reaction of Danishefsky's diene (1-Methoxy-3-trimethyl siloxy-1,3-butadiene, L14672) with imines in water under neutral conditions: Chem. Commun., 574 (2003).
- Lima, F. S.; Cuccovia, I. M.; Buchner, R.; Antunes, F. E.; Lindman, B.; Miguel, M. G.; Horinek, D.; Chaimovich, H. Sodium Triflate Decreases Interaggregate Repulsion and Induces Phase Separation in Cationic Micelles. Langmuir 2015, 31 (9), 2609-2614.
- Ji, X. M.; Wei, L.; Chen, F.; Tang, R. Y. Direct trifluoromethylation of imidazoheterocycles in a recyclable medium at room temperature. RSC Adv. 2015, 5 (38), 29766-29773.