Search
Thermo Scientific Chemicals
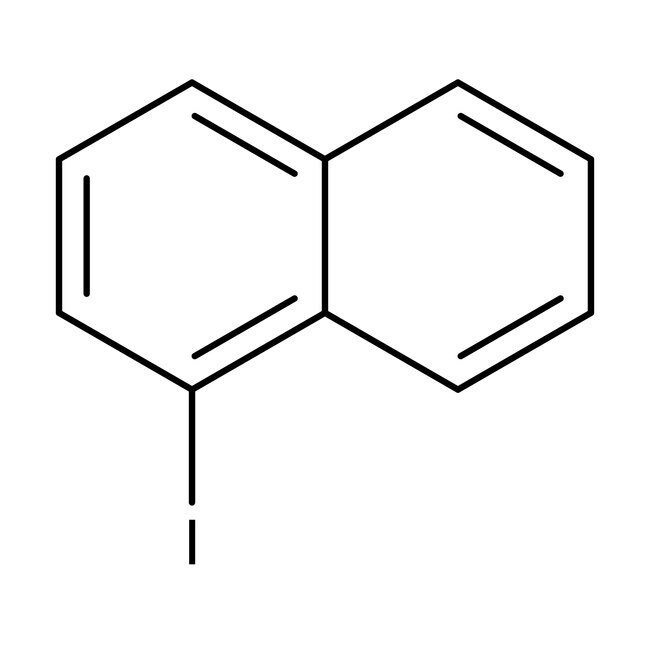
1-Yodonaftaleno, 98 %, Thermo Scientific Chemicals
CAS: 90-14-2 | C10H7I | 254.07 g/mol
Número de catálogo B22644.09
también denominado B22644-09
Precio (EUR)
69,30
Each
Cantidad:
10 g
Precio (EUR)
69,30
Each
Identificadores químicos
CAS90-14-2
IUPAC Name1-iodonaphthalene
Molecular FormulaC10H7I
InChI KeyNHPPIJMARIVBGU-UHFFFAOYSA-N
SMILESIC1=C2C=CC=CC2=CC=C1
Ver más
Especificaciones Hoja de especificaciones
Hoja de especificaciones
Appearance (Color)Clear yellow to brown to dark brown or purple
FormLiquid
Assay (GC)≥97.5%
Refractive Index1.6990-1.7030 @ 20°C
1-Iodonaphthalene undergoes Pd catalyzed cross-coupling reaction (Stille reaction) with 2,4-dimethoxy-6-(trimethylstannyl)pyrimidine to afford 2,4-dimethoxy-6-(naphthalen-1-yl)pyrimidine.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Aplicaciones
El 1-yodonaftaleno experimenta una reacción de acoplamiento cruzado catalizada por Pd (reacción de Stille) con 2,4-dimetoxi-6-(trimetilestannil)pirimidina que genera 2,4-dimetoxi-6-(naftalen-1-il)pirimidina.
Solubilidad
Ligeramente soluble en agua.
Notas
Sensible a la luz. Incompatible con agentes oxidantes.
El 1-yodonaftaleno experimenta una reacción de acoplamiento cruzado catalizada por Pd (reacción de Stille) con 2,4-dimetoxi-6-(trimetilestannil)pirimidina que genera 2,4-dimetoxi-6-(naftalen-1-il)pirimidina.
Solubilidad
Ligeramente soluble en agua.
Notas
Sensible a la luz. Incompatible con agentes oxidantes.
RUO – Research Use Only
General References:
- G.Kothandaraman; D.W.Pratt; D.S.Tinti. Optically detected magnetic resonance spectra of the lowest triplet state of 1-iodonaphthalene in durene at zero and high magnetic fields. J. Chem. Phys. 1975, 63,(3), 3337.
- Guillermo A.Lotz; Sara M.Palacios; Roberto A.Rossi. Stereoselective reaction of a chiral assisted amide enolate ion with 1-iodonaphthalene by the SRN1 mechanism. Tetrahedron Letters. 1994, 35,(42), 7711-7714.