Search
Thermo Scientific Chemicals
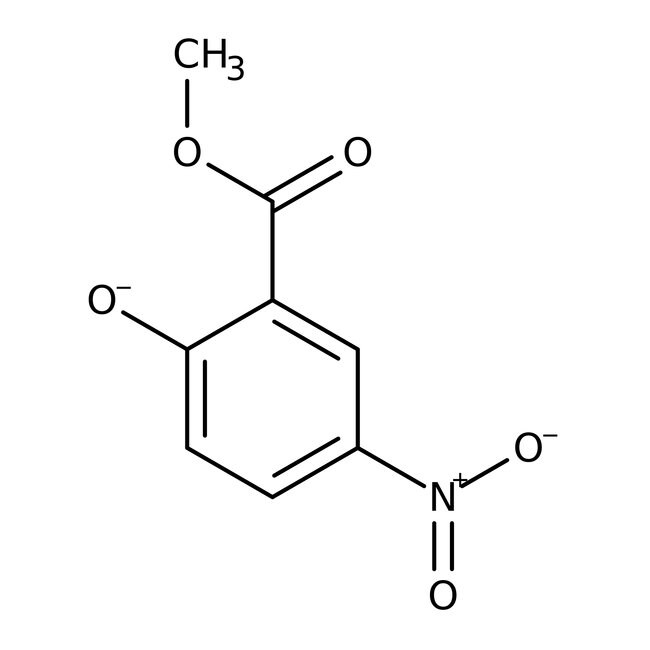
2-Hidroxi-5-nitrobenzoato de metilo, 98 %, Thermo Scientific Chemicals
CAS: 17302-46-4 | C8H7NO5 | 197.146 g/mol
Número de catálogo B25278.18
también denominado B25278-18
Precio (EUR)
135,00
Each
Cantidad:
50 g
Precio (EUR)
135,00
Each
Identificadores químicos
CAS17302-46-4
IUPAC Name2-(methoxycarbonyl)-4-nitrobenzen-1-olate
Molecular FormulaC8H6NO5
InChI KeyUUBFELFUKFJSRD-UHFFFAOYSA-M
SMILESCOC(=O)C1=CC(=CC=C1[O-])[N+]([O-])=O
Ver más
Especificaciones Hoja de especificaciones
Hoja de especificaciones
Appearance (Color)White to pale cream to cream to orange
Assay (GC)≥97.5%
Melting Point (clear melt)113.0-119.0?C
FormCrystals or powder or crystalline powder
Methyl 2-hydroxy-5-nitrobenzoate is used as pharmaceutical intermediate.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Aplicaciones
El 2-hidroxi-5-nitrobenzoato de metilo se utiliza como producto intermedio farmacéutico.
Solubilidad
Insoluble en agua.
Notas
Estable en las condiciones de almacenamiento recomendadas. Incompatible con agentes oxidantes.
El 2-hidroxi-5-nitrobenzoato de metilo se utiliza como producto intermedio farmacéutico.
Solubilidad
Insoluble en agua.
Notas
Estable en las condiciones de almacenamiento recomendadas. Incompatible con agentes oxidantes.
RUO – Research Use Only
General References:
- John McGinley; Vickie McKee; Hans Toftlund; John M.D.Walsh. Unusual metal coordination chemistry from an amino-amide derivative of 4-nitrophenol, a surprising ligand. Dalton Trans. 2009, (39), 8406-8412.
- David R.Edwards; Alexei A.Neverov; R.Stan Brown. Dissociative solvolytic cleavage of methyl (ortho-carboxymethyl)aryl phosphate diesters mediated by Yb3+ in methanol gives a 1012-fold rate acceleration attributable to leaving group assistance. J. Am. Chem. Soc. 2009, 131,(1), 368-377.