Search
Thermo Scientific Chemicals
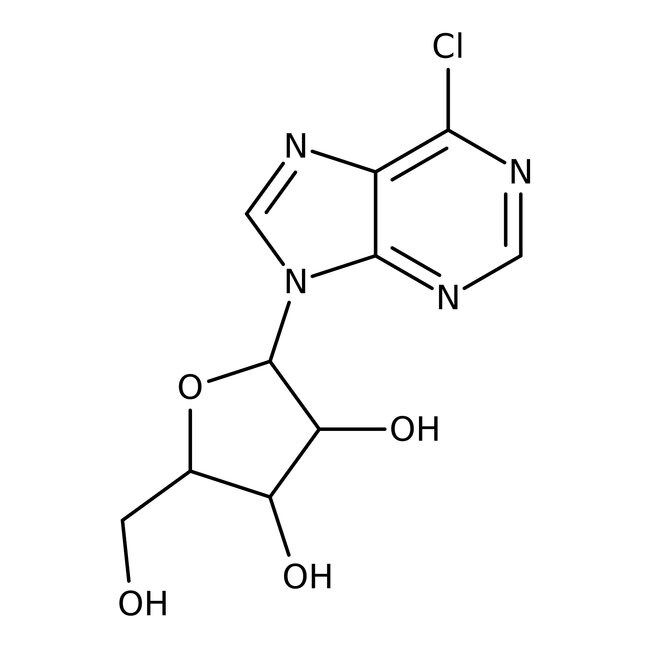
Ribósido de 6-cloropurina, 98 %, Thermo Scientific Chemicals
CAS: 5399-87-1 | C10H11ClN4O4 | 286.67 g/mol
Número de catálogo H59634.06
también denominado H59634-06
Precio (EUR)
453,00
Each
Cantidad:
5 g
Precio (EUR)
453,00
Each
Identificadores químicos
CAS5399-87-1
IUPAC Name2-(6-chloro-9H-purin-9-yl)-5-(hydroxymethyl)oxolane-3,4-diol
Molecular FormulaC10H11ClN4O4
InChI KeyXHRJGHCQQPETRH-UHFFFAOYNA-N
SMILESOCC1OC(C(O)C1O)N1C=NC2=C(Cl)N=CN=C12
Ver más
Especificaciones Hoja de especificaciones
Hoja de especificaciones
Assay (HPLC)>97.5%
6-Chloropurine riboside is used to study the kinetics and substrate specificity of adenosine deaminase. 6-Chloropurine riboside is benzoylated to facilitate synthesis of nucleoside derivatives such as 9-(2,3-Di-deoxy-2-fluoro-β-D-threo-pentofuranosyl)adenine. It , especially after phosphorylation to NMP, NDP or NTP, is used as a purine substrate analogue in studies with enzymes such as Inosine monophosphate dehydrogenase (IMPDH); bacteriophage T4 RNA-ligase (EC 6.5.1.3) and pancreatic fibonuclease A.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Aplicaciones
El ribósido de 6-cloropurina se utiliza para estudiar la cinética y la especificidad del sustrato de la adenosina deaminasa. El ribósido de 6-cloropurina está benzoilado para facilitar la síntesis de derivados de nucleósidos como la 9-(2,3-di-deoxi-2-fluoro-β-D-treo-pentofuranosil)adenina. Este, especialmente después de la fosforilación a NMP, NDP o NTP, se utiliza como análogo de sustrato de purina en estudios con enzimas como la inosina monofosfato deshidrogenasa (IMPDH), la ARN ligasa del bacteriófago T4 (EC 6.5.1.3) y la fibonucleasa pancreática A.
Solubilidad
Soluble en agua. (10,6 mg/ml).
Notas
Mantener alejado del calor y la humedad. Incompatible con agentes oxidantes fuertes.
El ribósido de 6-cloropurina se utiliza para estudiar la cinética y la especificidad del sustrato de la adenosina deaminasa. El ribósido de 6-cloropurina está benzoilado para facilitar la síntesis de derivados de nucleósidos como la 9-(2,3-di-deoxi-2-fluoro-β-D-treo-pentofuranosil)adenina. Este, especialmente después de la fosforilación a NMP, NDP o NTP, se utiliza como análogo de sustrato de purina en estudios con enzimas como la inosina monofosfato deshidrogenasa (IMPDH), la ARN ligasa del bacteriófago T4 (EC 6.5.1.3) y la fibonucleasa pancreática A.
Solubilidad
Soluble en agua. (10,6 mg/ml).
Notas
Mantener alejado del calor y la humedad. Incompatible con agentes oxidantes fuertes.
RUO – Research Use Only
General References:
- Juli Alonso.; M.Victòria Nogués.; Claudi M. Cuchillo. Modification of bovine pancreatic ribonuclease A with 6-chloropurine riboside. Arch. Biochem. Biophys. 1986, 246 (2),681-689.
- Michal Hocek .; Antonín Holý.; Ivan Votruba.; Hana Dvořáková. Synthesis and Cytostatic Activity of Substituted 6-Phenylpurine Bases and Nucleosides: Application of the Suzuki-Miyaura Cross-Coupling Reactions of 6-Chloropurine Derivatives with Phenylboronic Acids.J. Med. Chem. 2000, 43 (9),1817-1825.