Search
Thermo Scientific Chemicals
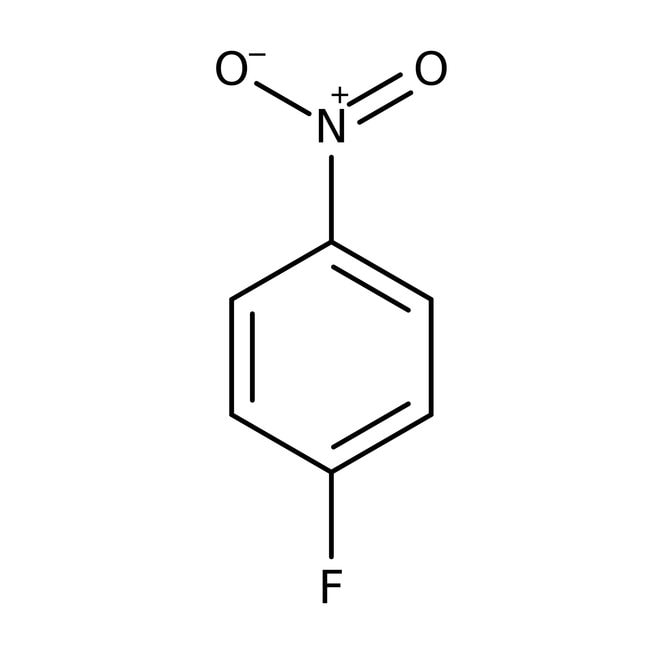
1-Fluoro-4-nitrobenzene, 99%
CAS: 350-46-9 | C6H4FNO2 | 141.101 g/mol
Catalog number A11057.36
also known as A11057-36
Price (EUR)
195,00
Each
Quantity:
500 g
Price (EUR)
195,00
Each
Chemical Identifiers
CAS350-46-9
IUPAC Name1-fluoro-4-nitrobenzene
Molecular FormulaC6H4FNO2
InChI KeyWFQDTOYDVUWQMS-UHFFFAOYSA-N
SMILES[O-][N+](=O)C1=CC=C(F)C=C1
View more
Specifications Specification Sheet
Specification Sheet
Assay (GC)> 98.5%
Formliquid as melt
Clarity25% in ethanol, clear solution
Refractive Index1.5300 - 1.5330 @20?C
Appearance (Color)Clear, yellow
1-Fluoro-4-nitrobenzene used as a component of hair dyes, as pharmaceutical intermediates. It is also used for the preparation of novel soluble aromatic polyimides
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
1-Fluoro-4-nitrobenzene used as a component of hair dyes, as pharmaceutical intermediates. It is also used for the preparation of novel soluble aromatic polyimides
Solubility
Insoluble in water.
Notes
Light Sensitive. Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Store away from oxidizing agents, acids, acid chlorides, acid anhydrides.
1-Fluoro-4-nitrobenzene used as a component of hair dyes, as pharmaceutical intermediates. It is also used for the preparation of novel soluble aromatic polyimides
Solubility
Insoluble in water.
Notes
Light Sensitive. Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Store away from oxidizing agents, acids, acid chlorides, acid anhydrides.
RUO – Research Use Only
General References:
- Shinji Takeda,; Hiromi Akiyama,; Hiroshi Kakiuchi. Synthesis and properties of bismaleimide resins containing ether bonds. Journal of Applied Polymer Science. 1988, 35 (5),1341-1350.
- William A. Feld,; B. Ramalingam,; Frank W. Harris. Polyimides containing oxyethylene units. Journal of Polymer Science: Polymer Chemistry Edition. 1983, 21(2),319-328,.
- On heating with potassium carbonate and silica at 200°, is converted to 4,4'-dinitrodiphenyl ether: J. Chem. Soc., Perkin 2, 377 (1992).