Search
Thermo Scientific Chemicals
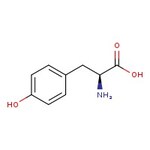
L-Tyrosine, 99%
CAS: 60-18-4 | C9H11NO3 | 181.19 g/mol
Catalog number A11141.22
also known as A11141-22
Price (EUR)
59,90
Each
Quantity:
100 g
Price (EUR)
59,90
Each
Chemical Identifiers
CAS60-18-4
IUPAC Name(2S)-2-amino-3-(4-hydroxyphenyl)propanoic acid
Molecular FormulaC9H11NO3
InChI KeyOUYCCCASQSFEME-SVGMAFHSNA-N
SMILESN[C@@H](CC1=CC=C(O)C=C1)C(O)=O
View more
Specifications Specification Sheet
Specification Sheet
Identification (FTIR)Conforms
FormPowder or crystalline powder
Optical Rotation-11? ? 1? (c= 4 in 1M HCl)
Appearance (Color)White
Assay (Non-aqueous acid-base Titration)≥98.5 to ≤101.5%
L-Tyrosine is used in pharmaceuticals, dietary supplements and food additives. It is a precursor to alkaloids such as morphine, neurotransmitters, epinephrine, p-coumaric acid, thyroxine, pigment melanin and catcholamines. It plays a vital role in photosynthesis.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
L-Tyrosine is used in pharmaceuticals, dietary supplements and food additives. It is a precursor to alkaloids such as morphine, neurotransmitters, epinephrine, p-coumaric acid, thyroxine, pigment melanin and catcholamines. It plays a vital role in photosynthesis.
Solubility
Soluble in hydrochloric acid. Slightly soluble in water.
Notes
Incompatible with strong oxidizing agents and strong reducing agents.
L-Tyrosine is used in pharmaceuticals, dietary supplements and food additives. It is a precursor to alkaloids such as morphine, neurotransmitters, epinephrine, p-coumaric acid, thyroxine, pigment melanin and catcholamines. It plays a vital role in photosynthesis.
Solubility
Soluble in hydrochloric acid. Slightly soluble in water.
Notes
Incompatible with strong oxidizing agents and strong reducing agents.
RUO – Research Use Only
General References:
- Gu, W.; Wang, M.; Mao, X.; Wang, Y.; Li, L.; Xia, W. A facile sensitive l-tyrosine electrochemical sensor based on a coupled CuO/Cu2O nanoparticles and multi-walled carbon nanotubes nanocomposite film. Anal. Methods 2015, 7 (4), 1313-1320.
- Hamzehzadeh, S.; Abbasi, M. The influence of 1-butyl-3-methyl-imidazolium bromide on the partitioning of l-tyrosine within the {polyethylene glycol 600+ potassium citrate} aqueous biphasic system at T= 298.15 K. J. Chem. Thermodyn. 2015, 80, 102-111.