Search
Thermo Scientific Chemicals
Esculin sesquihydrate, 97%
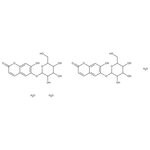
CAS: 66778-17-4 | C30H38O21 | 734.613 g/mol
Catalog number A11624.14
also known as A11624-14
Price (EUR)
139,00
Each
Quantity:
25 g
Price (EUR)
139,00
Each
Chemical Identifiers
CAS66778-17-4
IUPAC Namebis(7-hydroxy-6-{[3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy}-2H-chromen-2-one) trihydrate
Molecular FormulaC30H38O21
InChI KeyLGMHQDJZWHRZPO-UHFFFAOYNA-N
SMILESO.O.O.OCC1OC(OC2=C(O)C=C3OC(=O)C=CC3=C2)C(O)C(O)C1O.OCC1OC(OC2=C(O)C=C3OC(=O)C=CC3=C2)C(O)C(O)C1O
View more
Specifications Specification Sheet
Specification Sheet
Water Content (Karl Fischer Titration)5.03-9.57% (1-2 waters)
Optical Rotation-36.0° to -42.0° (c=5 in pyridine)
Appearance (Color)White to cream
FormPowder or crystalline powder
Assay (HPLC)≥96.0%
View more
Esculin sesquihydrate is used to inhibits chemically induced carcinogenesis in mouse skin and kidney, used in a microbiology laboratory to aid in the identification of bacterial species, a naturally occurring antioxidant.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Esculin sesquihydrate is used to inhibits chemically induced carcinogenesis in mouse skin and kidney, used in a microbiology laboratory to aid in the identification of bacterial species, a naturally occurring antioxidant.
Solubility
Slightly soluble in water.
Notes
Air Sensitive & Hygroscopic. Keep container tightly closed. Store in cool, dry conditions in well sealed containers.
Esculin sesquihydrate is used to inhibits chemically induced carcinogenesis in mouse skin and kidney, used in a microbiology laboratory to aid in the identification of bacterial species, a naturally occurring antioxidant.
Solubility
Slightly soluble in water.
Notes
Air Sensitive & Hygroscopic. Keep container tightly closed. Store in cool, dry conditions in well sealed containers.
RUO – Research Use Only
General References:
- Matthew C. Allendera.; Daniel B. Raudabaugh.; Frank H. Gleason.; Andrew N. Miller. The natural history, ecology, and epidemiology of Ophidiomyces ophiodiicola and its potential impact on free-ranging snake populations. Fungal Ecology. 2015, 17, 187-196.
- Cedric Tarayre.; Julien Bauwens.; Christel Matteotti.; Catherine Brasseur.; Catherine Millet.; Sebastien Massart.; Jacqueline Destain.; Micheline Vandenbol.; Edwin De Pauw.; Eric Haubruge.; Frederic Francis.; Philippe Thonart.; Daniel Portetelle.; Frank Delvigne. Multiple analyses of microbial communities applied to the gut of the wood-feeding termite Reticulitermes flavipes fed on artificial diets. Symbiosis. 2015, 65, (3), 143-155.
- Ultraviolet absorbent. Reversible inhibitor of phenylalanine hydroxylase.