Search
Thermo Scientific Chemicals
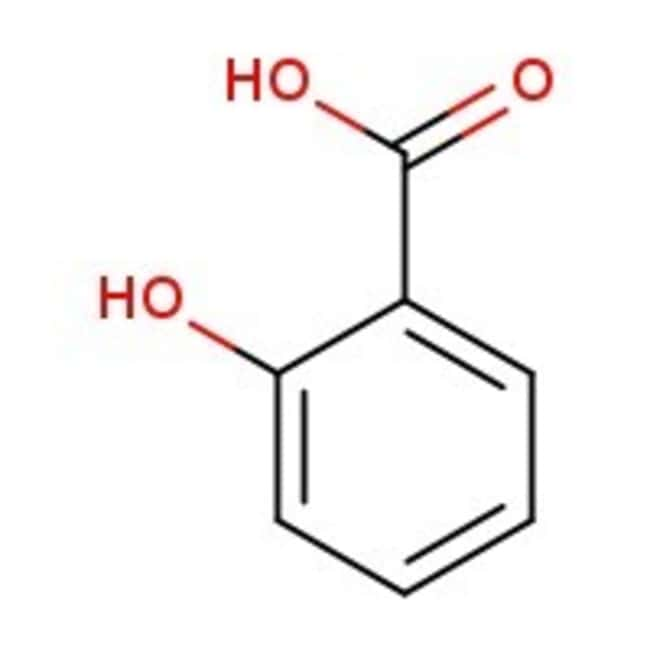
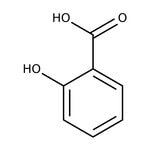
Salicylic acid, 99%
CAS: 69-72-7 | C7H6O3 | 138.12 g/mol
Catalog number A12253.30
also known as A12253-30
Price (EUR)
27,00
Each
Quantity:
250 g
Price (EUR)
27,00
Each
Chemical Identifiers
CAS69-72-7
IUPAC Name2-hydroxybenzoic acid
Molecular FormulaC7H6O3
InChI KeyYGSDEFSMJLZEOE-UHFFFAOYSA-N
SMILESOC(=O)C1=CC=CC=C1O
View more
Specifications Specification Sheet
Specification Sheet
Appearance (Color)White to cream
FormCrystals or powder or crystalline powder
Melting Point (clear melt)156-163?C
Assay (Aqueous acid-base Titration)≥98.5 to ≤101.5%
Assay (Silylated GC)≥98.5%
View more
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Suitable for battery materials development.
Applications
Salicylic acid is used as a food preservative as well as in the treatment of many skin disorders such as acne, dandruff, psoriasis and seborrheic dermatitis of the skin and scalp. It is an antiseptic. It is mainly used in the pharmaceutical industry for the manufacture of antipyretic, analgesic, anti-inflammatory and diuretic drugs. It is also used in dye industry for making azo direct dye and for acidic mordant dyeing.
Solubility
Soluble in carbon terachloride, benzene, propanol, absolute ethanol, acetone, toluene and ether. Slightly soluble in water.
Notes
Light sensitive. Incompatible with strong oxidizing agents, strong bases, iodine, iron and iron salts.
Salicylic acid is used as a food preservative as well as in the treatment of many skin disorders such as acne, dandruff, psoriasis and seborrheic dermatitis of the skin and scalp. It is an antiseptic. It is mainly used in the pharmaceutical industry for the manufacture of antipyretic, analgesic, anti-inflammatory and diuretic drugs. It is also used in dye industry for making azo direct dye and for acidic mordant dyeing.
Solubility
Soluble in carbon terachloride, benzene, propanol, absolute ethanol, acetone, toluene and ether. Slightly soluble in water.
Notes
Light sensitive. Incompatible with strong oxidizing agents, strong bases, iodine, iron and iron salts.
RUO – Research Use Only
General References:
- González-Ramos, K. M.; Fernández-Reyes, B.; Román, F. R.; Hernández-Maldonado, A. J. A hierarchical porous carbon-Mn+[FAU](Mn+ Ni2+ or Cu2+) adsorbent: Synthesis, characterization and adsorption of salicylic acid from water. Microporous Mesoporous Mater. 2014, 200, 225-234.
- Ouimet, M. A.; Fogaça, R.; Snyder, S. S.; Sathaye, S.; Catalani, L. H.; Pochan, D. J.; Uhrich, K. E. Poly (anhydride-ester) and Poly (N-vinyl-2-pyrrolidone) Blends: Salicylic Acid-Releasing Blends with Hydrogel-Like Properties that Reduce Inflammation. Macromol. Biosci. 2015, 15 (3), 342-350.