Search
Thermo Scientific Chemicals
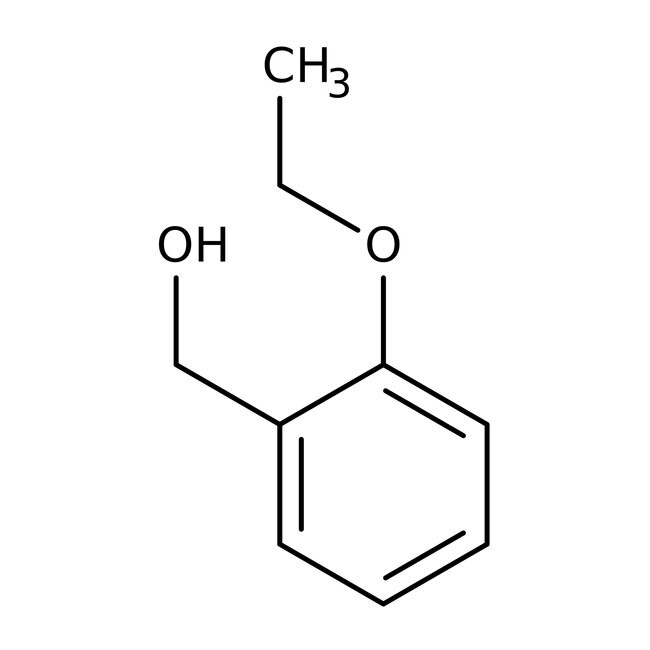
2-Ethoxybenzyl alcohol, 98%
CAS: 71672-75-8 | C9H12O2 | 152.19 g/mol
Catalog number A15477.22
also known as A15477-22
Price (EUR)
312,00
Each
Quantity:
100 g
Price (EUR)
312,00
Each
Chemical Identifiers
CAS71672-75-8
IUPAC Name(2-ethoxyphenyl)methanol
Molecular FormulaC9H12O2
InChI KeyICJVQAHPHKYCNU-UHFFFAOYSA-N
SMILESCCOC1=CC=CC=C1CO
View more
Specifications Specification Sheet
Specification Sheet
Assay (GC)≥97.5%
FormLiquid
Appearance (Color)Clear colorless to pale pink
Refractive Index1.5310-1.5350 @ 20?C
2-Ethoxybenzyl alcohol undergoes acetylation with acetic anhydride over zeolite to yield O-acetylated product. It is also used as pharmaceutical intermediates.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
2-Ethoxybenzyl alcohol undergoes acetylation with acetic anhydride over zeolite to yield O-acetylated product. It is also used as pharmaceutical intermediates.
Notes
Store in cool, dry conditions in a well sealed container. Store away from oxidizing agents.
2-Ethoxybenzyl alcohol undergoes acetylation with acetic anhydride over zeolite to yield O-acetylated product. It is also used as pharmaceutical intermediates.
Notes
Store in cool, dry conditions in a well sealed container. Store away from oxidizing agents.
RUO – Research Use Only
General References:
- Keith Smith; Gamal A el-Hiti; Anthony J Jayne; Michael Butters. Acetylation of aromatic ethers using acetic anhydride over solid acid catalysts in a solvent-free system. Scope of the reaction for substituted ethers. Organic & Biomolecular Chemistry. 2003, 1 (9), 1560-1564.
- D. E. Burton; K. Clarke and G. W. Gray. The mechanism of the antibacterial action of phenols and salicylaldehydes. Part III. Substituted benzaldehydes. J. Chem. Soc. 1964, 2458-2460.