Search
Thermo Scientific Chemicals
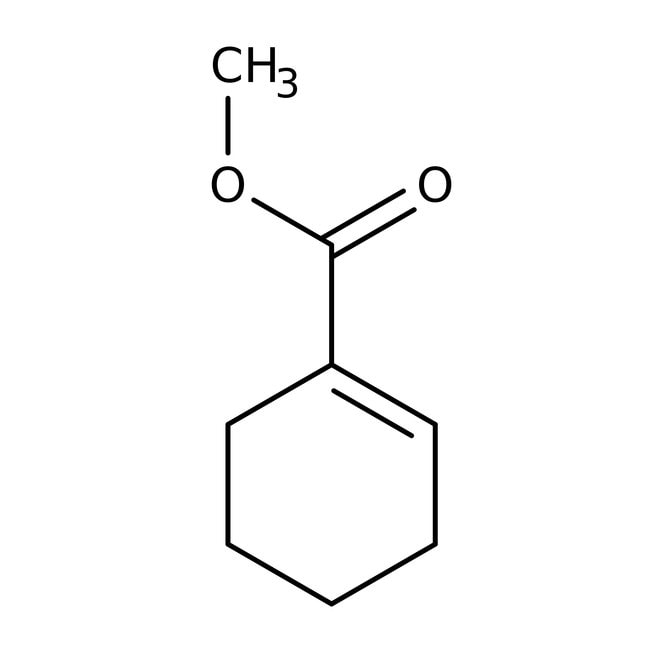
Methyl 1-cyclohexene-1-carboxylate, 97%
CAS: 18448-47-0 | C8H12O2 | 140.182 g/mol
Catalog number B25568.14
also known as B25568-14
Price (EUR)
392,00
Each
Quantity:
25 g
Price (EUR)
392,00
Each
Chemical Identifiers
CAS18448-47-0
IUPAC Namemethyl cyclohex-1-ene-1-carboxylate
Molecular FormulaC8H12O2
InChI KeyKXPWRCPEMHIZGU-UHFFFAOYSA-N
SMILESCOC(=O)C1=CCCCC1
View more
Specifications Specification Sheet
Specification Sheet
Appearance (Color)Clear colorless to pale yellow
Refractive Index1.4745-1.4795 @20?C
FormLiquid
Assay (GC)≥96.0%
Identification (FTIR)Conforms
Methyl 1-cyclohexene-1-carboxylate is used in the diastereoselective synthesis of cis-1,2-dialkenylcyclopropanols and methyl -7,7-dimethyl-9-oxo-1,3,4,4a,6,7,8,9,9b-decahydrodibenzo[b,d]furan-4a-carboxylate.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Methyl 1-cyclohexene-1-carboxylate is used in the diastereoselective synthesis of cis-1,2-dialkenylcyclopropanols and methyl -7,7-dimethyl-9-oxo-1,3,4,4a,6,7,8,9,9b-decahydrodibenzo[b,d]furan-4a-carboxylate.
Solubility
Immiscible with water.
Notes
Incompatible with strong oxidizing agents and bases.
Methyl 1-cyclohexene-1-carboxylate is used in the diastereoselective synthesis of cis-1,2-dialkenylcyclopropanols and methyl -7,7-dimethyl-9-oxo-1,3,4,4a,6,7,8,9,9b-decahydrodibenzo[b,d]furan-4a-carboxylate.
Solubility
Immiscible with water.
Notes
Incompatible with strong oxidizing agents and bases.
RUO – Research Use Only
General References:
- Johnson, M. R.; Gauuan, J. F.; Guo, C.; Guzzo, P. R.; Le, V. D.; Shenoy, R. A.; Hamby, J.; Roark, H.; Stier, M.; Mangette, J. E. Synthesis and Characterization of Novel Bi-and Tricyclic α-Amino Acids. Synth. Commun. 2011, 41 (18), 2769-2793.
- Tian, G. Q.; Yang, J.; Rosa-Perez, K. γ-Oxygenation of α, β-Unsaturated Esters by Vinylogous O-Nitroso Mukaiyama Aldol Reaction. Org. Lett. 2010, 12 (21), 5072-5074.