Search
Thermo Scientific Chemicals
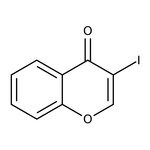
3-Iodochromone, 97%
CAS: 122775-34-2 | C9H5IO2 | 272.04 g/mol
Catalog number H34295.MD
also known as H34295-MD
Price (EUR)
107,00
Each
Quantity:
250 mg
Price (EUR)
107,00
Each
Chemical Identifiers
CAS122775-34-2
IUPAC Name3-iodo-4H-chromen-4-one
Molecular FormulaC9H5IO2
InChI KeySZDBDDKQJHHRGH-UHFFFAOYSA-N
SMILESIC1=COC2=CC=CC=C2C1=O
View more
Specifications Specification Sheet
Specification Sheet
FormCrystals or powder or crystalline powder
Assay (GC)≥96.0%
Melting Point (clear melt)92.0-101.0?C
Appearance (Color)Pale yellow
3-Iodochromone is used in the synthesis of 2-(N-azolyl)chromones, 3-azolylthiochromones.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
3-Iodochromone is used in the synthesis of 2-(N-azolyl)chromones, 3-azolylthiochromones.
Notes
Light sensitive. Store in the dark. Store away from oxidizing agent, light.
3-Iodochromone is used in the synthesis of 2-(N-azolyl)chromones, 3-azolylthiochromones.
Notes
Light sensitive. Store in the dark. Store away from oxidizing agent, light.
RUO – Research Use Only
General References:
- Yoshiaki Sugita, Ichiro Yokoe. Reaction of 3-iodochromone with nucleophile 1. Reaction of 3-iodochromone with azoles. Heterocycles. 1996, 43 (11), 2503-2511.
- Ichiro Hayakawa; Akiyuki Ikedo; Hideo Kigoshi. Synthesis of Glaziovianin A: A Potent Antitumor Isoflavone. Chemistry Letters. 2007, 36 (11), 1382-1383.