Search
Thermo Scientific Chemicals
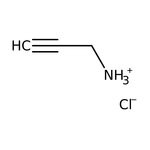
Propargylamine, 98%
CAS: 2450-71-7 | C3H5N | 55.08 g/mol
Price (EUR)
68,40
Each
Chemical Identifiers
CAS2450-71-7
IUPAC Nameprop-2-yn-1-aminium chloride
Molecular FormulaC3H6ClN
InChI KeyIKXNIQJDNKPPCH-UHFFFAOYSA-N
SMILES[Cl-].[NH3+]CC#C
View more
Specifications Specification Sheet
Specification Sheet
Assay (GC)>97.5
Propargylamine is used in the synthesis of a chiral, a fluorescent macrocycle by 1,3-dipolar cycloaddition of propargyl amides of carbohydrate-linked amino acids and 9,10-bis(azidomethyl)anthracene. It acts as an intermediate in the production of drugs.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Propargylamine is used in the synthesis of a chiral, a fluorescent macrocycle by 1,3-dipolar cycloaddition of propargyl amides of carbohydrate-linked amino acids and 9,10-bis(azidomethyl)anthracene. It acts as an intermediate in the production of drugs.
Solubility
Miscible with water, chloroform and ethyl acetate.
Notes
Hygroscopic. Light sensitive. Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Incompatible with acids, acid chlorides, acid anhydrides, strong oxidizing agents and carbon dioxide.
Propargylamine is used in the synthesis of a chiral, a fluorescent macrocycle by 1,3-dipolar cycloaddition of propargyl amides of carbohydrate-linked amino acids and 9,10-bis(azidomethyl)anthracene. It acts as an intermediate in the production of drugs.
Solubility
Miscible with water, chloroform and ethyl acetate.
Notes
Hygroscopic. Light sensitive. Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Incompatible with acids, acid chlorides, acid anhydrides, strong oxidizing agents and carbon dioxide.
RUO – Research Use Only
General References:
- He, Y.; Liang, Y.; Wang, D. The highly sensitive and facile colorimetric detection of the glycidyl azide polymer based on propargylamine functionalized gold nanoparticles using click chemistry. Chem. Commun. 2015, 51 (60), 12092-12094.
- Zindo, F.; Joubert, J.; Malan, S. Polycyclic propargylamine derivatives as multifunctional neuroprotective agents: cum laude. S Afr. Pharm. J. 2015, 82 (2), 32-34.