Search
Thermo Scientific Chemicals
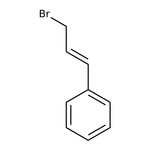
Cinnamyl bromide, predominantly trans, 95%
CAS: 4392-24-9 | C9H9Br | 197.075 g/mol
Catalog number L01420.22
also known as L01420-22
Price (EUR)
258,00
Each
Quantity:
100 g
Price (EUR)
258,00
Each
Chemical Identifiers
CAS4392-24-9
IUPAC Name[(1E)-3-bromoprop-1-en-1-yl]benzene
Molecular FormulaC9H9Br
InChI KeyRUROFEVDCUGKHD-QPJJXVBHSA-N
SMILESBrC\C=C\C1=CC=CC=C1
View more
Specifications Specification Sheet
Specification Sheet
FormCrystals or powder or crystalline powder or fused solid or clear liquid as melt
Appearance (Color)White to pale cream to cream to yellow to orange to dark brown to brown
Assay (GC)≥94.0%
Refractive Index1.6160-1.6220 @ 20?C
Cinnamyl bromide, is used in the synthesis of naftifine.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Cinnamyl bromide, is used in the synthesis of naftifine.
Solubility
Soluble in methanol. Insoluble in water.
Notes
Light Sensitive. Keep container tightly closed. Store away from oxidizing agents.
Cinnamyl bromide, is used in the synthesis of naftifine.
Solubility
Soluble in methanol. Insoluble in water.
Notes
Light Sensitive. Keep container tightly closed. Store away from oxidizing agents.
RUO – Research Use Only
General References:
- C.B. Kanner.; U.K. Pandit. Functionalized enamines—XXXII: Reaction of β-amino-α, β-unsaturated esters and amides with benzyl and cinnamyl bromides. Tetrahedron. 1982, 38, (24), 3597-3604.
- Methvin B.Isaac.; Tak-Hang Chan. Indium mediated coupling of aldehydes with allyl bromides in aqueous media. the issue of regio- and diastereo-selectivity. Tetrahedron Letters. 1995, 36, (49), 8957-8960.