Search
Alfa Aesar
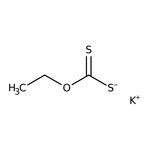
Potassium ethyl xanthate, 97+%
CAS: 140-89-6 | C3H5KOS2 | 160.29 g/mol
Catalog number ALF-A11450-22
HSN 29309099
Price (INR)
5,520.00
Each
Quantity:
100 g
Price (INR)
5,520.00
Each
Chemical Identifiers
CAS140-89-6
IUPAC Namepotassium (ethoxymethanethioyl)sulfanide
Molecular FormulaC3H5KOS2
InChI KeyJCBJVAJGLKENNC-UHFFFAOYSA-M
SMILES[K+].CCOC([S-])=S
View more
Specifications Specification Sheet
Specification Sheet
Appearance (Color)Pale cream to cream to yellow
FormCrystals or powder or crystalline powder
Assay (Iodometric Titration)≥97.5 to ≤102.5% (Dry wt. basis)
Loss on Drying≤2% (105°C/constant weight)
Identification (FTIR)Conforms
Potassium ethyl xanthate is used as a reagent for extraction of metal ions into organic solvents and in the preparation of 2-mercapto benzimidazoles and 2-mercapto benzoxazoles by reacting with o-phenylenediamines and o-amino phenols respectively. It acts as a precursor in the production of unsymmetrical sulfides through cross-coupling reaction and as a floatation agent in the mining industry. It is involved in the determination of cadmium in water samples by substoichiometric radiochemical method and the adsorption of colloidal dye disperse Blue 3 onto pretreated polyester fabric.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Potassium ethyl xanthate is used as a reagent for extraction of metal ions into organic solvents and in the preparation of 2-mercapto benzimidazoles and 2-mercapto benzoxazoles by reacting with o-phenylenediamines and o-amino phenols respectively. It acts as a precursor in the production of unsymmetrical sulfides through cross-coupling reaction and as a floatation agent in the mining industry. It is involved in the determination of cadmium in water samples by substoichiometric radiochemical method and the adsorption of colloidal dye disperse Blue 3 onto pretreated polyester fabric.
Solubility
Soluble in water.
Notes
Light sensitive. Incompatible with strong acids, strong bases and strong oxidizing agents and water.
Potassium ethyl xanthate is used as a reagent for extraction of metal ions into organic solvents and in the preparation of 2-mercapto benzimidazoles and 2-mercapto benzoxazoles by reacting with o-phenylenediamines and o-amino phenols respectively. It acts as a precursor in the production of unsymmetrical sulfides through cross-coupling reaction and as a floatation agent in the mining industry. It is involved in the determination of cadmium in water samples by substoichiometric radiochemical method and the adsorption of colloidal dye disperse Blue 3 onto pretreated polyester fabric.
Solubility
Soluble in water.
Notes
Light sensitive. Incompatible with strong acids, strong bases and strong oxidizing agents and water.
RUO – Research Use Only
General References:
- Reagent for extraction of metal ions into organic solvents: Talanta, 23, 417 (1975).
- For conversion of arene diazonium salts to thiols via the aryl ethyl xanthate, see: Org. Synth. Coll., 3, 809 (1955). [Caution! explosive diazonium xanthates may be formed: Org. Synth. Coll., 5, 1050 (1973)].
- For use in the conversion of o-phenylenediamines to 2-mercapto benzimidazoles and o-amino phenols to 2-mercapto benzoxazoles, see: Org. Synth. Coll., 4, 569 (1963).
- Reaction with the tosylate of a secondary alcohol, followed by cleavage with ethylenediamine gives the thiol of opposite configuration: Synthesis, 425 (1974).
- Mahmoud, M. R.; Lazaridis, N. K.; Matis, K. A. Study of flotation conditions for cadmium(II) removal from aqueous solutions. Process Saf. Environ. Prot. 2015, 94, 203-211.
- Deng, J.; Lei, Y.; Wen, S.; Chen, Z. Modeling interactions between ethyl xanthate and Cu/Fe ions using DFT/B3LYP approach. Int. J. Miner. Process. 2015, 140, 43-49.