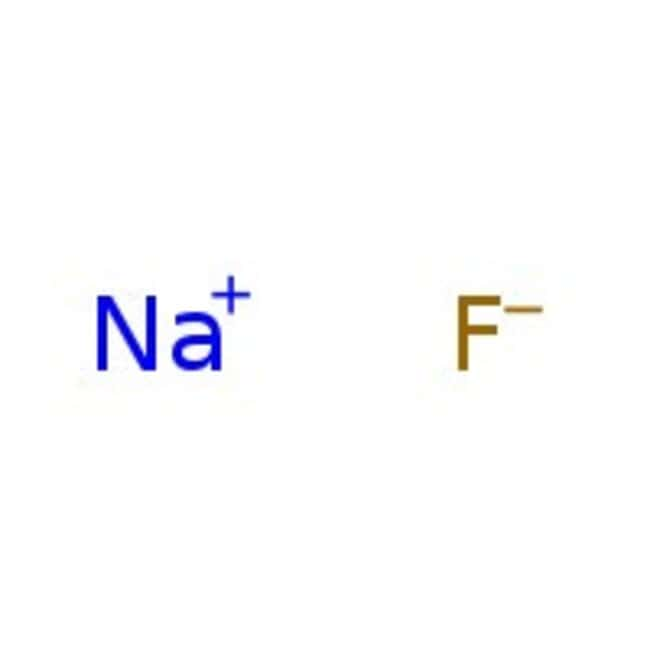Search
Alfa Aesar
Sodium fluoride, 99%, Thermo Scientific Chemicals
Catalog number ALF-A13019-0I
HSN 28261990
Price (INR)
27,211.00
Each
Quantity:
5000 g
Price (INR)
27,211.00
Each
Chemical Identifiers
CAS7681-49-4
IUPAC Namesodium fluoride
Molecular FormulaFNa
InChI KeyPUZPDOWCWNUUKD-UHFFFAOYSA-M
SMILES[F-].[Na+]
View more
Specifications Specification Sheet
Specification Sheet
Appearance (Color)White
Assay from Supplier's CofA≥98.5 to ≤101.5%
Loss on Drying<0.5% (150?C/constant wt)
FormPowder
Sodium fluoride is used as a steel degreasing agent and in tin electroplating, flux, heat treating salt compositions, fluoridation of drinking water, removing HF from exhaust systems to reduce air pollution, and frosting glasses. It serves as a source of fluoride ion in several applications including it use in synthetic chemistry, for example in Finkelstein reaction for preparating fluoro compounds, desilylation in sensitive organic compounds, metallurgy, and reaction with electrophilic chlorides. It has medical and other applications including complexing of magnesium ions.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Sodium fluoride is used as a steel degreasing agent and in tin electroplating, flux, heat treating salt compositions, fluoridation of drinking water, removing HF from exhaust systems to reduce air pollution, and frosting glasses. It serves as a source of fluoride ion in several applications including it use in synthetic chemistry, for example in Finkelstein reaction for preparating fluoro compounds, desilylation in sensitive organic compounds, metallurgy, and reaction with electrophilic chlorides. It has medical and other applications including complexing of magnesium ions.
Solubility
Slightly soluble in water. Insoluble in alcohol
Notes
It must be kept away from oxidizing agents, metals, acids, alkalis. It is less hygroscopic.
Sodium fluoride is used as a steel degreasing agent and in tin electroplating, flux, heat treating salt compositions, fluoridation of drinking water, removing HF from exhaust systems to reduce air pollution, and frosting glasses. It serves as a source of fluoride ion in several applications including it use in synthetic chemistry, for example in Finkelstein reaction for preparating fluoro compounds, desilylation in sensitive organic compounds, metallurgy, and reaction with electrophilic chlorides. It has medical and other applications including complexing of magnesium ions.
Solubility
Slightly soluble in water. Insoluble in alcohol
Notes
It must be kept away from oxidizing agents, metals, acids, alkalis. It is less hygroscopic.
RUO – Research Use Only
General References:
- Reviews: Fluoride ion as a base in organic synthesis: Chem. Rev., 80, 429 (1980). Alkali metal fluorides in organic synthesis: Synthesis, 169 (1983). See also Potassium fluoride, 14130.
- Songsiripradubboon, S.; Hamba, H.; Trairatvorakul, C.; Tagami, J. Sodium fluoride mouthrinse used twice daily increased incipient caries lesion remineralization in an in situ model. J. Dentistry 2014, 42 (3), 271-278.
- Atmaca, N.; Atmaca, H. T.; Kanicic, A.; Anteplioglu, T. Protective effect of resveratrol on sodium fluoride-induced oxidative stress, hepatotoxicity and neurotoxicity in rats. Food Chem. Toxicol. 2014, 70, 191-197.