Search
Thermo Scientific Chemicals
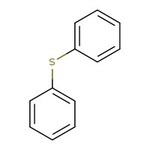
Diphenyl sulfide, 98%
CAS: 139-66-2 | C12H10S | 186.27 g/mol
Catalog number A11114.22
also known as A11114-22
Price (TWD)
-
Quantity:
100 g
Chemical Identifiers
CAS139-66-2
IUPAC Name(phenylsulfanyl)benzene
Molecular FormulaC12H10S
InChI KeyLTYMSROWYAPPGB-UHFFFAOYSA-N
SMILESS(C1=CC=CC=C1)C1=CC=CC=C1
View more
Specifications Specification Sheet
Specification Sheet
FormLiquid
Refractive Index1.6310-1.6350 @ 20?C
Identification (FTIR)Conforms
Appearance (Color)Clear colorless
Assay (GC)≥97.5%
Diphenyl sulfide is used as a raw material for triaryl sulfonium photoinitiator, intermediates for agrichemicals, catalyst, metal extractants.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Diphenyl sulfide is used as a raw material for triaryl sulfonium photoinitiator, intermediates for agrichemicals, catalyst, metal extractants.
Solubility
Soluble in ether, benzene, carbon disulphide.
Notes
Light Sensitive. Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Store away from oxidizing agents, acids, acid chlorides, acid anhydrides.
Diphenyl sulfide is used as a raw material for triaryl sulfonium photoinitiator, intermediates for agrichemicals, catalyst, metal extractants.
Solubility
Soluble in ether, benzene, carbon disulphide.
Notes
Light Sensitive. Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Store away from oxidizing agents, acids, acid chlorides, acid anhydrides.
RUO – Research Use Only
General References:
- Kenji. Uneyama,; Sigeru. Torii. Electrochemistry of organic sulfur compounds. III. Novel anodic synthesis of a sulfonium salt from diphenyl sulfide. J. Org. Chem. 1972, 37(3),367-369.
- J. Eriksen,; C. S. Foote,; T. L. Parker. Photosensitized oxygenation of alkenes and sulfides via a non-singlet-oxygen mechanism. J. Am. Chem. Soc 1977, 99 (19), 6455-6456.