Search
Thermo Scientific Chemicals
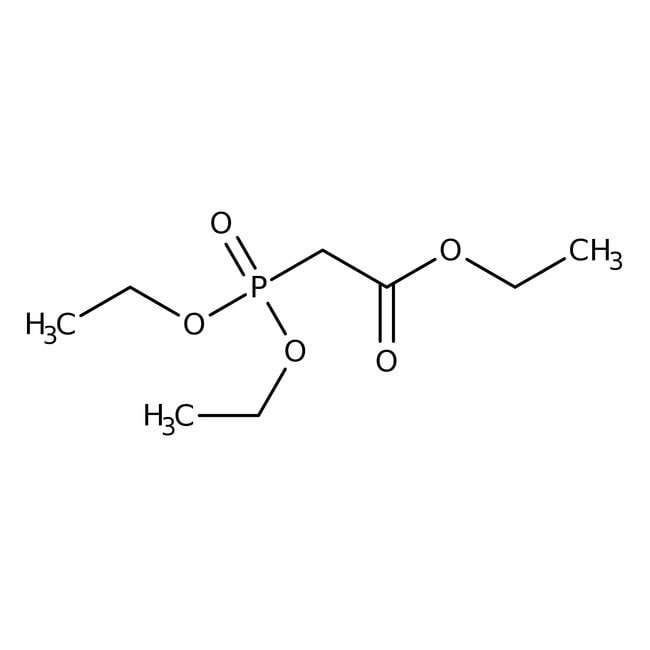
Triethyl phosphonoacetate, 98+%
CAS: 867-13-0 | C8H17O5P | 224.193 g/mol
Catalog number A14120.36
also known as A14120-36
Price (TWD)
-
Quantity:
500 g
Chemical Identifiers
CAS867-13-0
IUPAC Nameethyl 2-(diethoxyphosphoryl)acetate
Molecular FormulaC8H17O5P
InChI KeyGGUBFICZYGKNTD-UHFFFAOYSA-N
SMILESCCOC(=O)CP(=O)(OCC)OCC
View more
Specifications Specification Sheet
Specification Sheet
Assay (GC)≥98.0%
Refractive Index1.4300-1.4340 @ 20?C
Appearance (Color)Clear colorless
Identification (FTIR)Conforms
FormLiquid
Triethyl phosphonoacetate serves as a reactant used in Horner-Wadsworth-Emmons reactions, Tsuji-Trost type reactions, Intramolecular Heck-type cyclization and isomerizations and Intramolecular aryne-ene reactions. In Horner-Wadsworth-Emmons reaction, it is utilized as a reagent to prepare chiral 2-methylcyclopropanecarboxylic acid from (S)-propylene oxide.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Triethyl phosphonoacetate serves as a reactant used in Horner-Wadsworth-Emmons reactions, Tsuji-Trost type reactions, Intramolecular Heck-type cyclization and isomerizations and Intramolecular aryne-ene reactions. In Horner-Wadsworth-Emmons reaction, it is utilized as a reagent to prepare chiral 2-methylcyclopropanecarboxylic acid from (S)-propylene oxide.
Solubility
Slightly miscible with water.
Notes
Incompatible with strong oxidizing agents.
Triethyl phosphonoacetate serves as a reactant used in Horner-Wadsworth-Emmons reactions, Tsuji-Trost type reactions, Intramolecular Heck-type cyclization and isomerizations and Intramolecular aryne-ene reactions. In Horner-Wadsworth-Emmons reaction, it is utilized as a reagent to prepare chiral 2-methylcyclopropanecarboxylic acid from (S)-propylene oxide.
Solubility
Slightly miscible with water.
Notes
Incompatible with strong oxidizing agents.
RUO – Research Use Only
General References:
- Wadsworth-Emmons (Horner) reaction with aldehydes and ketones gives acrylic esters. See, e.g.: Org. Synth. Coll., 5, 509, 547 (1973), and Appendix 1.
- Base-catalyzed aldol, Cannizzaro and saponification reactions can be minimized, by the use of, e.g.: K2CO3: Synthesis, 300 (1983); Tetrahedron Lett., 26, 53 (1985); F-: Tetrahedron Lett., 21, 2161 (1980); N-ethyldiisopropylamine + LiCl: Tetrahedron Lett., 25, 2183 (1984); Et3N + Li halides: J. Org. Chem., 50, 2624 (1985).
- Reaction with aqueous formaldehyde does not lead to the expected Wadsworth-Emmons product, but to ethyl ɑ-(hydroxymethyl)acrylate, a precursor of the synthetically useful ethyl ɑ-bromomethylacrylate, by condensation with the initially-formed aldol product: Org. Synth. Coll., 8, 265 (1993).
- Kim, H.; Paik, Y. K. Synthesis of Photoaffinity-Labeled Daumone Analogs. Bull. Korean Chem. Soc. 2015, 36 (9), 2177-2178.
- Lee, J. Y.; Choi, J. H.; Ryoo, K. S.; Kwon, Y. B.; Hong, Y. P. Economical Synthesis of Grapevine Moth Sex Pheromone. Bull. Korean Chem. Soc. 2015, 36 (1), 421-423.