Search
Thermo Scientific Chemicals
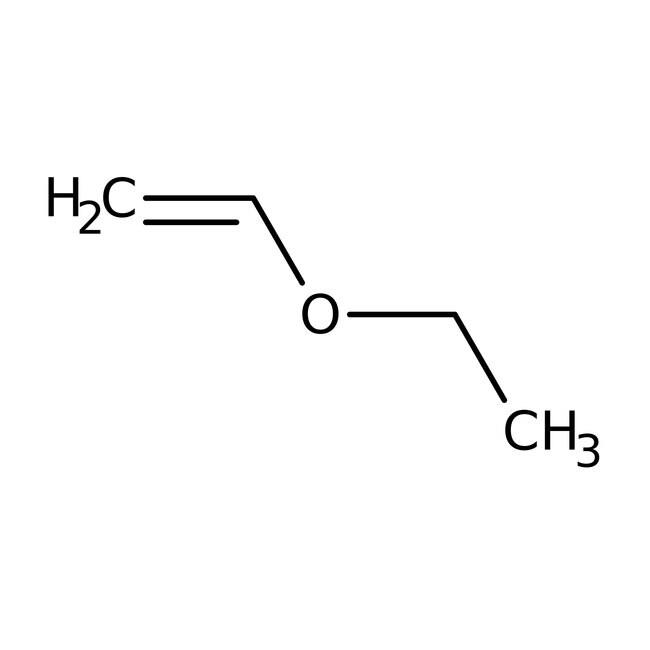
Ethyl vinyl ether, 99%, stab.
CAS: 109-92-2 | C4H8O | 72.11 g/mol
Catalog number A15691.0F
also known as A15691-0F
Price (TWD)
-
Quantity:
2500 mL
Chemical Identifiers
CAS109-92-2
IUPAC Nameethoxyethene
Molecular FormulaC4H8O
InChI KeyFJKIXWOMBXYWOQ-UHFFFAOYSA-N
SMILESCCOC=C
View more
Specifications Specification Sheet
Specification Sheet
Appearance (Color)Clear, colourless
Formliquid
Assay (GC)> 98.5%
Refractive Index1.3755-1.3785
Ethyl vinyl ether is used in fragrances, lubricating oil additives and spices preparation. It acts as an intermediate for sulfadiazine. It finds application in pharmaceuticals as anesthetics and analgesics. Further, it is used as a solvent in organic synthesis.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Ethyl vinyl ether is used in fragrances, lubricating oil additives and spices preparation. It acts as an intermediate for sulfadiazine. It finds application in pharmaceuticals as anesthetics and analgesics. Further, it is used as a solvent in organic synthesis.
Solubility
Miscible with common organic solvents. Slightly miscible with water.
Notes
Air sensitive. Incompatible with strong oxidizing agents, oxygen and acids. Store in a cool place.
Ethyl vinyl ether is used in fragrances, lubricating oil additives and spices preparation. It acts as an intermediate for sulfadiazine. It finds application in pharmaceuticals as anesthetics and analgesics. Further, it is used as a solvent in organic synthesis.
Solubility
Miscible with common organic solvents. Slightly miscible with water.
Notes
Air sensitive. Incompatible with strong oxidizing agents, oxygen and acids. Store in a cool place.
RUO – Research Use Only
General References:
- Acid-catalyzed reaction with alcohols gives 1-ethoxyethyl (EE) ethers, useful as protected derivatives, e.g. in reactions with organometallics. Possible catalysts include: tosic acid: J. Am. Chem. Soc., 101, 7104 (1979); Org. Synth., 76, 199 (1998), PPTS: Tetrahedron Lett., 28, 4303 (1987), or dichloroacetic acid: J. Org. Chem., 37, 1947 (1972). Cleavage of this mixed acetal occurs in dilute acid, more readily than THP ether (see 3,4-Dihydro-2H-pyran, L02731). Protection of cyanohydrins allows use as acyl anion equivalents: Org. Synth. Coll., 7, 381 (1990):
- Also used in the protection of phenols as EE ethers which can be cleaved by acid-catalyzed methanolysis: J. Am. Chem. Soc., 119, 8071 (1997).
- In the presence of Montmorillonite K10, an addition reaction with diethyl acetals gives the ß-ethoxy acetal of the bis-homologated aldehyde, providing, on acid cleavage, an effective route to the corresponding ɑß-unsaturated aldehyde: Synthesis, 137 (1981).
- Khavani, M.; Izadyar, M. A comprehensive study of the solvent effects on the cycloaddition reaction of diethyl azodicarboxylate and ethyl vinyl ether: Efficient implementation of QM and TD-DFT study. Int. J. Quantum Chem. 2015, 115 (6), 381-388.
- Han, D.; Cao, H.; Li, M.; Li, X.; Zhang, S.; He, M.; Hu, J. Computational study on the mechanisms and rate constants of the OH-initiated oxidation of ethyl vinyl ether in atmosphere. J. Phys. Chem. A 2015, 119 (4), 719-727.