Search
Thermo Scientific Chemicals
Sodium tetraborate decahydrate, 99+%
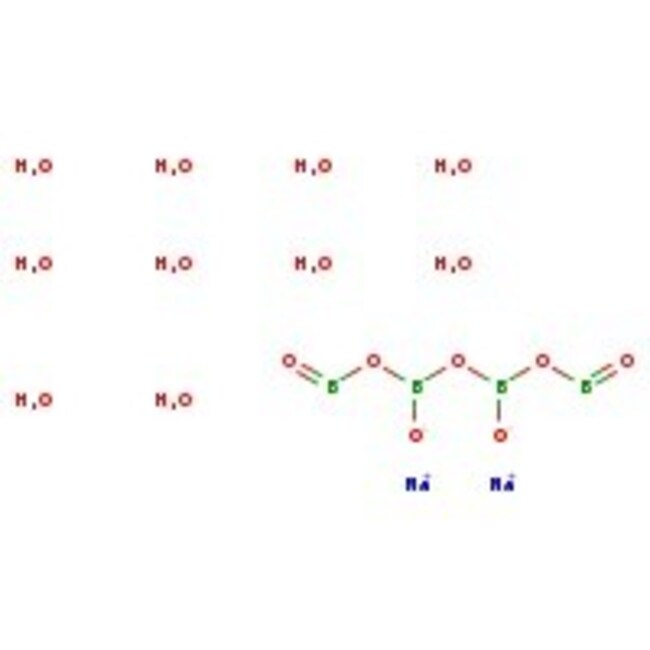
CAS: 1303-96-4 | B4Na2O7·10H2O
Catalog number A16176.0C
also known as A16176-0C
Price (TWD)
-
Quantity:
10,000 g
Chemical Identifiers
CAS1303-96-4
IUPAC Namedisodium decahydrate oxoboranyl ({oxido[(oxoboranyl)oxy]boranyl}oxy)boronate
Molecular FormulaB4H20Na2O17
InChI KeyOJPFLODMGUZMKD-UHFFFAOYSA-N
SMILESO.O.O.O.O.O.O.O.O.O.[Na+].[Na+].[O-]B(OB=O)OB([O-])OB=O
View more
Specifications Specification Sheet
Specification Sheet
FormCrystals or powder or crystalline powder
Identification (FTIR)Conforms
Appearance (Color)White
Assay (Aqueous acid-base Titration)≥99.0 to ≤105.0%
It is used in detergents, cosmetics, and enamel glazes. It is likewise employed to prepare buffer solutions in biochemistry, as a fire retardant, anti-fungal compound, in the manufacture of fiberglass, as a flux in metallurgy, neutron-capture shields for radioactive sources, a texturing agent in cooking, and as a harbinger for other boron compounds. The molten borax bath is useful for carbide coating process on metals. It is applied as an efficient metal-free catalyst for Hetero-Michael reactions in an aqueous medium.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
It is used in detergents, cosmetics, and enamel glazes. It is likewise employed to prepare buffer solutions in biochemistry, as a fire retardant, anti-fungal compound, in the manufacture of fiberglass, as a flux in metallurgy, neutron-capture shields for radioactive sources, a texturing agent in cooking, and as a harbinger for other boron compounds. The molten borax bath is useful for carbide coating process on metals. It is applied as an efficient metal-free catalyst for Hetero-Michael reactions in an aqueous medium.
Solubility
Soluble in water, glycerol and ethanol. Insoluble in acids
Notes
Incompatible with strong oxidizing agents and strong acids.
It is used in detergents, cosmetics, and enamel glazes. It is likewise employed to prepare buffer solutions in biochemistry, as a fire retardant, anti-fungal compound, in the manufacture of fiberglass, as a flux in metallurgy, neutron-capture shields for radioactive sources, a texturing agent in cooking, and as a harbinger for other boron compounds. The molten borax bath is useful for carbide coating process on metals. It is applied as an efficient metal-free catalyst for Hetero-Michael reactions in an aqueous medium.
Solubility
Soluble in water, glycerol and ethanol. Insoluble in acids
Notes
Incompatible with strong oxidizing agents and strong acids.
RUO – Research Use Only
General References:
- Nasir, M. Z. M.; Sofer, Z.; Ambrosi, A.; Pumera, M. A limited anodic and cathodic potential window of MoS2: limitations in electrochemical applications. Nanoscale 2015, 7 (7), 3126-3129.
- Hussain, S.; Bharadwaj, S. K.; Chaudhuri, M. K.; Kalita, H. Borax as an Efficient Metal-Free Catalyst for Hetero-Michael Reactions in an Aqueous Medium. Eur. J. org. Chem. 2007, 2007 (2), 374-378.