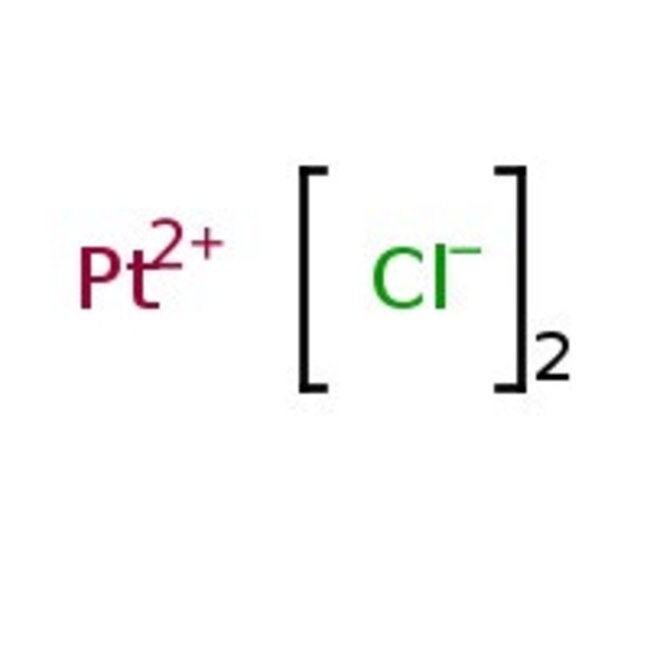Search
Thermo Scientific Chemicals
Platinum(II) chloride, 98%
CAS: 10025-65-7 | Cl2Pt | 265.98 g/mol
Chemical Identifiers
CAS10025-65-7
IUPAC Nameplatinum(2+) dichloride
Molecular FormulaCl2Pt
InChI KeyCLSUSRZJUQMOHH-UHFFFAOYSA-L
SMILES[Cl-].[Cl-].[Pt++]
View more
Specifications Specification Sheet
Specification Sheet
FormPowder or Crystals
Elemental Analysis(Platinum) : 71.5-75.1%
Assay (unspecified)(by Platinum EA) : >97.5 to <102.5%
Appearance (Color)Green to brown
Platinum(II) chloride is used as a catalyst for a variety of C-C bond, C-O bond, and C-N bond formation reactions. It acts as a precursor to prepare other platinum compounds such as cisplatin and dichloro(cycloocta-1,5-diene)platinum(II).
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Platinum(II) chloride is used as a catalyst for a variety of C-C bond, C-O bond, and C-N bond formation reactions. It acts as a precursor to prepare other platinum compounds such as cisplatin and dichloro(cycloocta-1,5-diene)platinum(II).
Solubility
Soluble in ammonia and hydrochloric acid. Insoluble in water, alcohol and ether.
Notes
Incompatible with strong oxidizing agents.
Platinum(II) chloride is used as a catalyst for a variety of C-C bond, C-O bond, and C-N bond formation reactions. It acts as a precursor to prepare other platinum compounds such as cisplatin and dichloro(cycloocta-1,5-diene)platinum(II).
Solubility
Soluble in ammonia and hydrochloric acid. Insoluble in water, alcohol and ether.
Notes
Incompatible with strong oxidizing agents.
RUO – Research Use Only
General References:
- Sivaraman, M.; Perumal, P. T. Synthesis of tetracyclic chromenones via platinum(ii) chloride catalysed cascade cyclization of enediyne-enones. Org. Biomol. Chem. 2014, 12 (8), 1318-1327.
- Schelwies, M.; Farwick, A.; Rominger, F.; Helmchen, G. Platinum(II) Chloride-Catalyzed Stereoselective Domino Enyne Isomerization/Diels- Alder Reaction. J. Org. Chem. 2010, 75 (22), 7917-7919.