Search
Thermo Scientific Chemicals
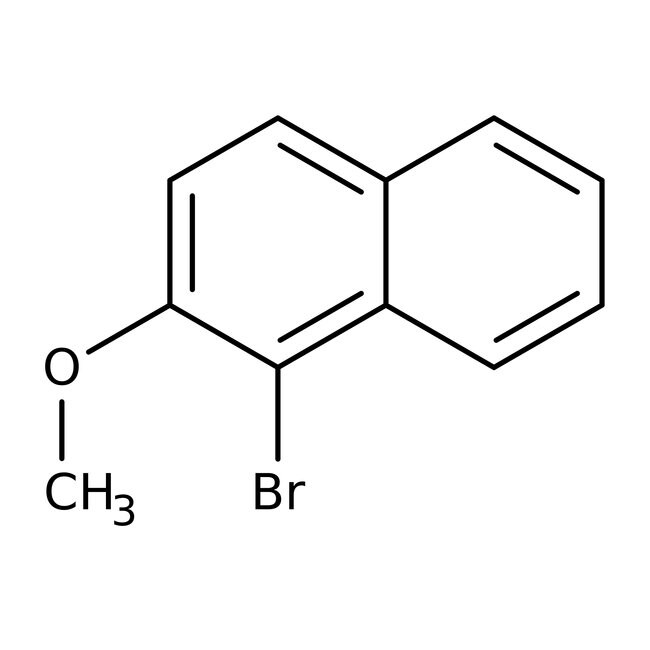
1-Bromo-2-methoxynaphthalene, 97%
CAS: 3401-47-6 | C11H9BrO | 237.10 g/mol
Catalog number A18926.22
also known as A18926-22
Price (TWD)
-
Quantity:
100 g
Chemical Identifiers
CAS3401-47-6
IUPAC Name1-bromo-2-methoxynaphthalene
Molecular FormulaC11H9BrO
InChI KeyXNIGURFWNPLWJM-UHFFFAOYSA-N
SMILESCOC1=CC=C2C=CC=CC2=C1Br
View more
Specifications Specification Sheet
Specification Sheet
Appearance (Color)White
FormCrystals or powder or crystalline powder
Melting Point79.5-88.5?C
Assay (GC)≥96.0%
1-Bromo-2-methoxy-naphthalene is a useful synthetic intermediate. It is used as a reagent to synthesize a catalyst for highly enantioselective aziridination of styrene derivatives. It can also be used to prepare biaryls or biheterocycles by palladium-catalyzed Ullmann coupling.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
1-Bromo-2-methoxy-naphthalene is a useful synthetic intermediate. It is used as a reagent to synthesize a catalyst for highly enantioselective aziridination of styrene derivatives. It can also be used to prepare biaryls or biheterocycles by palladium-catalyzed Ullmann coupling.
Solubility
Slightly soluble in water.
Notes
Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Store away from oxidizing agent.
1-Bromo-2-methoxy-naphthalene is a useful synthetic intermediate. It is used as a reagent to synthesize a catalyst for highly enantioselective aziridination of styrene derivatives. It can also be used to prepare biaryls or biheterocycles by palladium-catalyzed Ullmann coupling.
Solubility
Slightly soluble in water.
Notes
Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Store away from oxidizing agent.
RUO – Research Use Only
General References:
- M. Carmen Carreno; Jose L. Garcia Ruano; Gema Sanz; Miguel A. Toledo; Antonio Urbano. N-Bromosuccinimide in Acetonitrile: A Mild and Regiospecific Nuclear Brominating Reagent for Methoxybenzenes and Naphthalenes. J. Org. Chem., 1995, 60 (16), 5328-5331.
- George Majetich; Rodgers Hicks; and Steven Reister. Electrophilic Aromatic Bromination Using Bromodimethylsulfonium Bromide Generated in Situ. J. Org. Chem, 1997, 62(13), 4321-4326.