Search
Thermo Scientific Chemicals
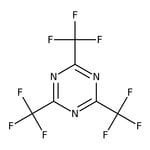
2,4,6-Tris(trifluoromethyl)-1,3,5-triazine, 98%
CAS: 368-66-1 | C6F9N3 | 285.07 g/mol
Specifications
Beilstein302339
CAS368-66-1
Chemical Name or Material2,4,6-Tris(trifluoromethyl)-1,3,5-triazine
DOT InformationHazard Class: 6.1; Packaging Group: II
Density1.596
View more
2,4,6-Tris(trifluoromethyl)-1,3,5-triazine is used to prepare 1-phenyl-4,6-bis-trifluoromethyl-1H-pyrazolo[3,4-d]pyrimidine by reacting with sodium 5-amino-1-phenyl-1H-pyrazole-4-carboxylate.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
2,4,6-Tris(trifluoromethyl)-1,3,5-triazine is used to prepare 1-phenyl-4,6-bis-trifluoromethyl-1H-pyrazolo[3,4-d]pyrimidine by reacting with sodium 5-amino-1-phenyl-1H-pyrazole-4-carboxylate.
Notes
Air and moisture sensitive. Store in a cool place. Keep the container tightly closed in a dry and well-ventilated place. Incompatible with strong acids, strong bases, strong oxidizing agents and strong reducing agents.
2,4,6-Tris(trifluoromethyl)-1,3,5-triazine is used to prepare 1-phenyl-4,6-bis-trifluoromethyl-1H-pyrazolo[3,4-d]pyrimidine by reacting with sodium 5-amino-1-phenyl-1H-pyrazole-4-carboxylate.
Notes
Air and moisture sensitive. Store in a cool place. Keep the container tightly closed in a dry and well-ventilated place. Incompatible with strong acids, strong bases, strong oxidizing agents and strong reducing agents.
RUO – Research Use Only
General References:
- Huang, Z.; Pan, Y.; Chao, Y.; Shen, W.; Wang, C.; Xu, H. Triazaheterocyclic compound as an efficient catalyst for dehydration of fructose into 5-hydroxymethylfurfural. RSC Adv. 2014, 4 (26), 13434-13437.
- Rosa, M. D.; Arnold, D.; Hartline, D. Four Mechanisms in the Reactions of 3-Aminopyrrole with 1,3,5-Triazines: Inverse Electron Demand Diels-Alder Cycloadditions vs SNAr Reactions via Uncatalyzed and Acid-Catalyzed Pathways. J. Org. Chem. 2013, 78 (17), 8614-8623.