Search
Thermo Scientific Chemicals
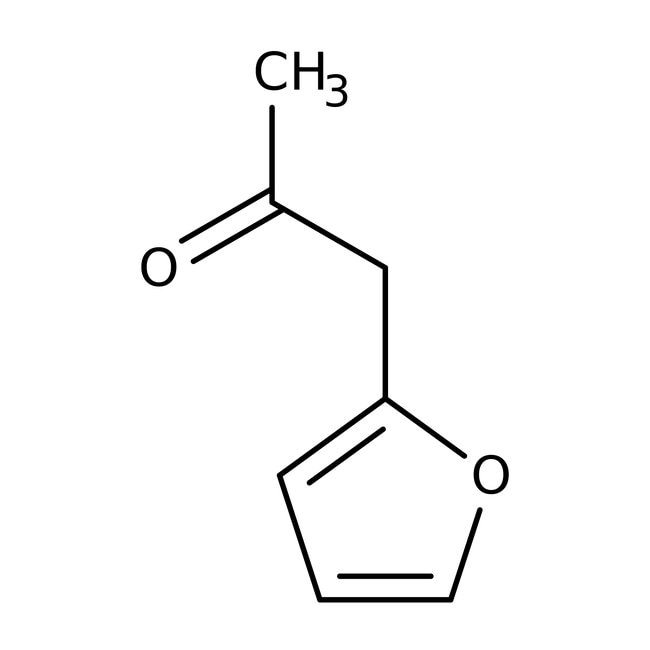
2-Furylacetone, 99%
CAS: 6975-60-6 | C7H8O2 | 124.139 g/mol
Catalog number B22171.14
also known as B22171-14
Price (TWD)
-
Quantity:
25 g
Chemical Identifiers
CAS6975-60-6
IUPAC Name1-(furan-2-yl)propan-2-one
Molecular FormulaC7H8O2
InChI KeyIQOJTGSBENZIOL-UHFFFAOYSA-N
SMILESCC(=O)CC1=CC=CO1
View more
Specifications Specification Sheet
Specification Sheet
Appearance (Color)Clear colorless to yellow
Assay (GC)≥98.5%
Refractive Index1.4680-1.4735 @ 20?C
FormLiquid
Identification (FTIR)Conforms
Suitable for fragrance applications.2-Furylacetone is used as the reactant in the synthesis and transformations of furan derivatives
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Suitable for fragrance applications.2-Furylacetone is used as the reactant in the synthesis and transformations of furan derivatives
Solubility
Soluble in alcohol, triacetin.
Notes
Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Store away from strong oxidizing agents.
Suitable for fragrance applications.2-Furylacetone is used as the reactant in the synthesis and transformations of furan derivatives
Solubility
Soluble in alcohol, triacetin.
Notes
Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Store away from strong oxidizing agents.
RUO – Research Use Only
General References:
- Hiroshi Yamaoka,; Ikuhiro Mishima,; Mitsuko Miyamoto,; Terukiyo Hanafusa. The Acid-catalyzed Reaction of the 2-Oxabicyclo[4.1.0]hept-3-en-5-one System: Isomerization from Homo-4-pyrones into 2-Furylacetone Derivatives. Bulletin of the Chemical Society of Japan. 1980, 53 (2), 469-477.
- A. Ya. Medne,;N. O. Saldabol. Synthesis and transformations of furan derivatives. Chemistry of Heterocyclic Compounds. 1966, 1 (4), 423-425.