Search
Thermo Scientific Chemicals
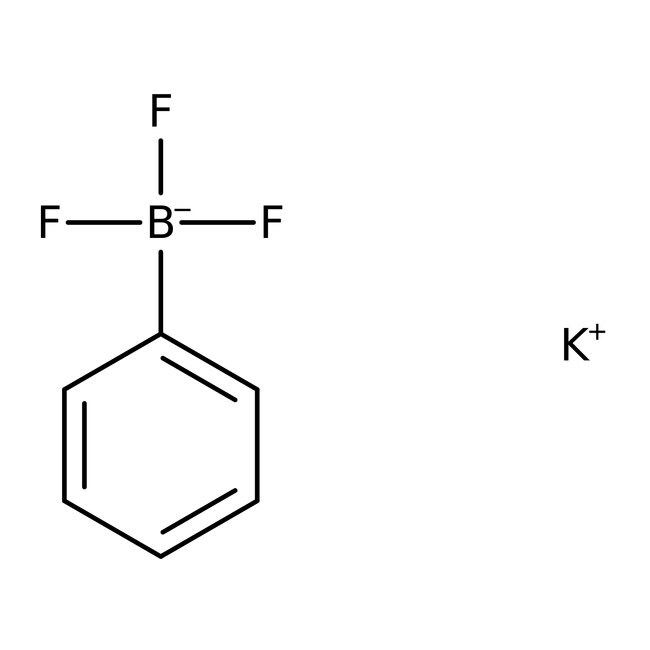
Potassium phenyltrifluoroborate, 98%
CAS: 153766-81-5 | C6H5BF3K | 184.01 g/mol
Chemical Identifiers
CAS153766-81-5
IUPAC Namepotassium trifluoro(phenyl)boranuide
Molecular FormulaC6H5BF3K
InChI KeyDVAFPKUGAUFBTJ-UHFFFAOYSA-N
SMILES[K+].F[B-](F)(F)C1=CC=CC=C1
View more
Specifications Specification Sheet
Specification Sheet
Appearance (Color)White to cream
FormCrystalline powder or flake
Assay (unspecified)≥97.5% by ¹H-NMR
Identification (FTIR)Conforms
Aryltrifluoroborate salts are more nucleophilic than the corresponding arylboronic acids and undergo ligand-free Pd-catalyzed cross-coupling reactions with arenediazonium tetrafluoroborates to give good yields of biaryls. Suzuki Cross-Coupling are conducted using organotrifluoroborates as a potent boronic acid surrogates. In the presence of Dicarbonyl(2,4-pentanedionato)rhodium(I), 39295, K aryl- and alkenyltrifluoroborates add to aldehydes and enones to give secondary alcohols and saturated ketones, respectively.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Aryltrifluoroborate salts are more nucleophilic than the corresponding arylboronic acids and undergo ligand-free Pd-catalyzed cross-coupling reactions with arenediazonium tetrafluoroborates to give good yields of biaryls. Suzuki Cross-Coupling are conducted using organotrifluoroborates as a potent boronic acid surrogates. In the presence of Dicarbonyl(2,4-pentanedionato)rhodium(I), 39295, K aryl- and alkenyltrifluoroborates add to aldehydes and enones to give secondary alcohols and saturated ketones, respectively.
Solubility
Soluble in water.
Notes
Store in cool dry conditions in well sealed containers. Incompatible with strong oxidizing agents.
Aryltrifluoroborate salts are more nucleophilic than the corresponding arylboronic acids and undergo ligand-free Pd-catalyzed cross-coupling reactions with arenediazonium tetrafluoroborates to give good yields of biaryls. Suzuki Cross-Coupling are conducted using organotrifluoroborates as a potent boronic acid surrogates. In the presence of Dicarbonyl(2,4-pentanedionato)rhodium(I), 39295, K aryl- and alkenyltrifluoroborates add to aldehydes and enones to give secondary alcohols and saturated ketones, respectively.
Solubility
Soluble in water.
Notes
Store in cool dry conditions in well sealed containers. Incompatible with strong oxidizing agents.
RUO – Research Use Only
General References:
- Alexander K.L. Yuen.; Craig A. Hutton. Deprotection of pinacolyl boronate esters via hydrolysis of intermediate potassium trifluoroborates.Tetrahedron Lett. 2005, 46 (46),7899-7903 .
- George W. Kabalk.; Mohammad Al-Masum. Microwave enhanced cross-coupling reactions involving potassium organotrifluoroborates.Tetrahedron Lett. 2005, 46 (37),6329-6331 .
- Treatment with TMS chloride in acetonitrile generates the Lewis acid phenylboron difluoride in situ, avoiding the need to handle boron trihalides: J. Org. Chem., 60, 3020 (1995).
- Aryltrifluoroborate salts are more nucleophilic than the corresponding arylboronic acids and undergo ligand-free Pd-catalyzed cross-coupling reactions with arenediazonium tetrafluoroborates to give good yields of biaryls: Tetrahedron Lett., 38, 4393 (1997); Eur. J. Org. Chem., 1875 (1999). Cross-coupling with aryl or heteroaryl halides and triflates, under ligand-free conditions, has subsequently been reported: Org. Lett., 4, 1867 (2002).
- Coupling with diaryliodonium salts has also been reported: Synth. Commun., 29, 2457 (1999). The method has been extended to the synthesis of benzophenones by carbonylative cross-coupling in the presence of CO: J. Chem. Res. (Synop.), 400 (1999).
- In the presence of Dicarbonyl (2,4-pentanedionato) rhodium(I) , 39295, K aryl- and alkenyltrifluoroborates add to aldehydes and enones to give secondary alcohols and saturated ketones, respectively: Org. Lett., 1, 1683 (1999):
- Asymmetric Rh-catalyzed additions to enones in the presence of a chiral BINAP catalyst give the corresponding saturated ketones in high yield and ee: Eur. J. Org. Chem., 3552 (2002).