Search
Thermo Scientific Chemicals
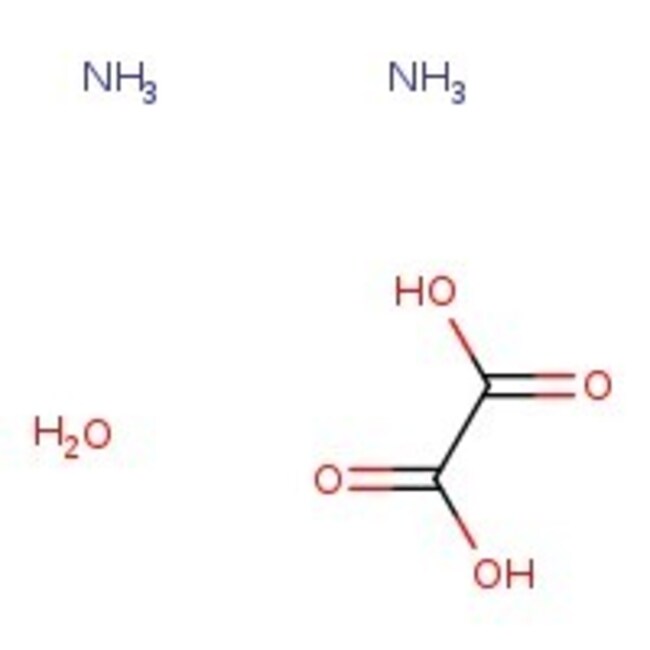
Ammonium oxalate monohydrate, 98%
CAS: 6009-70-7 | C2H10N2O5 | 142.111 g/mol
化學識別
CAS6009-70-7
IUPAC Nameoxalic acid diamine hydrate
Molecular FormulaC2H10N2O5
InChI KeyMSMNVXKYCPHLLN-UHFFFAOYSA-N
SMILESN.N.O.OC(=O)C(O)=O
檢視更多
規格 規格表
規格表
Assay (Non-aqueous acid-base Titration)≥97.5 to ≤102.5%
Appearance (Color)White to pale cream
Loss on Drying≤20.0% (60°C/constant wt.)
FormCrystals or powder or crystalline powder
Identification (FTIR)Conforms
Ammonium oxalate monohydrate is widely utilized as a buffering reagent and a dispersant to determine the surface interaction of platelets. It finds an application to study its acute poisoning effect on sheep and to investigate the formation of oxalate monoalkylamide in the human lens. It is also used in the detection and determination of calcium, lead, fluoride and rare earth metals. It is employed as chelators and forms complexes with metals. It acts as reducing agent in gold extraction and is an active ingredient of ferrous metal surface polishing fluid. It is a promoting agent in production of cobalt oxide and nickel oxide.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Ammonium oxalate monohydrate is widely utilized as a buffering reagent and a dispersant to determine the surface interaction of platelets. It finds an application to study its acute poisoning effect on sheep and to investigate the formation of oxalate monoalkylamide in the human lens. It is also used in the detection and determination of calcium, lead, fluoride and rare earth metals. It is employed as chelators and forms complexes with metals. It acts as reducing agent in gold extraction and is an active ingredient of ferrous metal surface polishing fluid. It is a promoting agent in production of cobalt oxide and nickel oxide.
Solubility
Soluble in water. Slightly soluble in alcohol.
Notes
Incompatible with strong oxidizing agents and strong acids.
Ammonium oxalate monohydrate is widely utilized as a buffering reagent and a dispersant to determine the surface interaction of platelets. It finds an application to study its acute poisoning effect on sheep and to investigate the formation of oxalate monoalkylamide in the human lens. It is also used in the detection and determination of calcium, lead, fluoride and rare earth metals. It is employed as chelators and forms complexes with metals. It acts as reducing agent in gold extraction and is an active ingredient of ferrous metal surface polishing fluid. It is a promoting agent in production of cobalt oxide and nickel oxide.
Solubility
Soluble in water. Slightly soluble in alcohol.
Notes
Incompatible with strong oxidizing agents and strong acids.
RUO – Research Use Only
General References:
- Kripal, R.; Yadav, A. K. Theoretical calculation of zero field splitting parameters of Cr3+doped ammonium oxalate monohydrate. Physica B. 2015, 466, 16-18.
- Qiao, Y.; Wang, K.; Yuan, H.; Yang, K.; Zou, B. Negative Linear Compressibility in Organic Mineral Ammonium Oxalate Monohydrate with Hydrogen Bonding Wine-Rack Motifs. J. Phys. Chem. Lett. 2015, 6 (14), 2755-2760.