Search
Thermo Scientific Chemicals
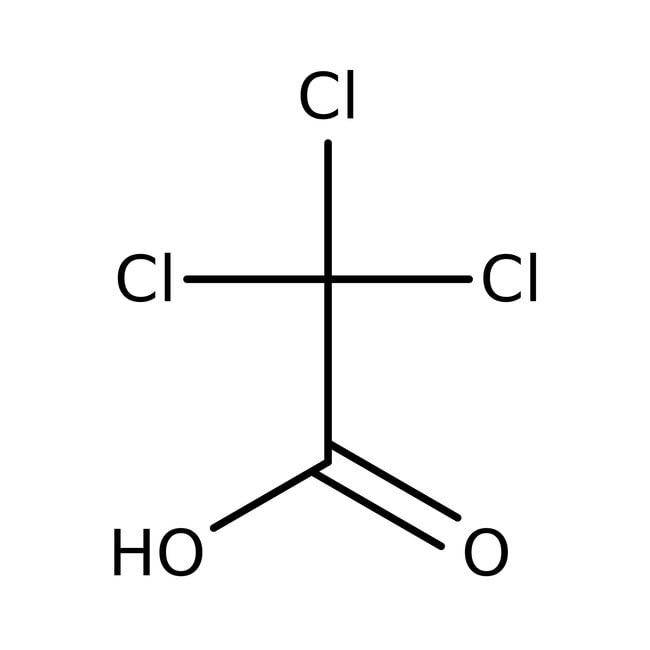
Trichloroacetic acid, 99%
CAS: 76-03-9 | C2HCl3O2 | 163.378 g/mol
化學識別
CAS76-03-9
IUPAC Nametrichloroacetic acid
Molecular FormulaC2HCl3O2
InChI KeyYNJBWRMUSHSURL-UHFFFAOYSA-N
SMILESOC(=O)C(Cl)(Cl)Cl
檢視更多
規格 規格表
規格表
Identification (FTIR)Conforms
Assay (Aqueous acid-base Titration)≥98.5 to ≤101.5%
Water Content (Karl Fischer Titration)<0.5%
Appearance (Color)White to pale yellow
Melting Point (clear melt)52-60?C
檢視更多
Protein precipitation reagent Trichloroacetic acid is used as a precipitating agent in biochemistry for precipitation of proteins, DNA and RNA. It is an active ingredient used for cosmetic treatments like chemical peels, tattoo removal and the treatment of warts including genital warts. It is also used to determine protein concentration and a decalcifier and fixative in microscopy.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Protein precipitation reagent Trichloroacetic acid is used as a precipitating agent in biochemistry for precipitation of proteins, DNA and RNA. It is an active ingredient used for cosmetic treatments like chemical peels, tattoo removal and the treatment of warts including genital warts. It is also used to determine protein concentration and a decalcifier and fixative in microscopy.
Solubility
Soluble in water, alcohol, ethyl ether, benzene, acetone, o-xylene and carbon tetrachloride.
Notes
Moisture sensitive. Hygroscopic. Store in cool place. Incompatible with strong oxidizing agents, strong bases and amines.
Protein precipitation reagent Trichloroacetic acid is used as a precipitating agent in biochemistry for precipitation of proteins, DNA and RNA. It is an active ingredient used for cosmetic treatments like chemical peels, tattoo removal and the treatment of warts including genital warts. It is also used to determine protein concentration and a decalcifier and fixative in microscopy.
Solubility
Soluble in water, alcohol, ethyl ether, benzene, acetone, o-xylene and carbon tetrachloride.
Notes
Moisture sensitive. Hygroscopic. Store in cool place. Incompatible with strong oxidizing agents, strong bases and amines.
RUO – Research Use Only
General References:
- Trihaloacetic acids react with aldehydes in DMSO at room temperature to give trihalomethyl carbinols: J. Chem. Soc., Perkin 2, 1247 (1984):
- The acid and its Na salt (1:1) in DMF are also effective: Tetrahedron Lett., 33, 3435 (1992). Use of HMPA or DMI extends the scope to less reactive aldehydes: Synthesis, 327 (1990).
- Malachová, A.; Štočková, L.; Wakker, A.; Varga, E.; Krska, R.; Michlmayr, H.; Adam, G.; Berthiller, F. Critical evaluation of indirect methods for the determination of deoxynivalenol and its conjugated forms in cereals. Anal. Bioanal. Chem. 2015, 407 (20), 6009-6020.
- Young, M. S.; van Tran, K.; Goh, E.; Shia, J. C. A Rapid SPE-Based Analytical Method for UPLC/MS/MS Determination of Aminoglycoside Antibiotic Residues in Bovine Milk, Muscle, and Kidney. J. AOAC Int. 2014, 97 (6), 1737-1741.