Search
Thermo Scientific Chemicals
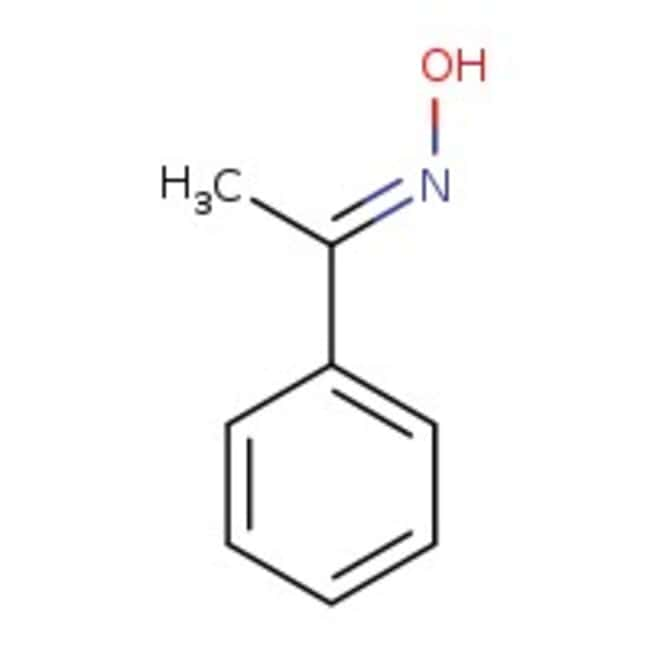
Acetophenone oxime, 98%
CAS: 613-91-2 | C8H9NO | 135.17 g/mol
化學識別
CAS613-91-2
IUPAC Name(E)-N-(1-phenylethylidene)hydroxylamine
Molecular FormulaC8H9NO
InChI KeyJHNRZXQVBKRYKN-VQHVLOKHSA-N
SMILESC\C(=N/O)C1=CC=CC=C1
檢視更多
規格 規格表
規格表
Appearance (Color)White to cream
Formcrystalline powder
Assay (GC)> 97.5%
Clarity2.5% w/v solution in ethanol will be transparent to almost transparent by JIS K8001 5.2
Used as a ligand in transition-metal complex catalyst chemistry. Oxime acts as an antioxidant, radical scavenger which find applications in textile, plastic, paint, detergent, and rubber industry.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Used as a ligand in transition-metal complex catalyst chemistry. Oxime acts as an antioxidant, radical scavenger which find applications in textile, plastic, paint, detergent, and rubber industry.
Solubility
Slightly soluble in water. Soluble in ethyl alcohol almost transparency.
Notes
Air and Moisture Sensitive. Store away from air and water/moisture. Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Store away from oxidizing agent.
Used as a ligand in transition-metal complex catalyst chemistry. Oxime acts as an antioxidant, radical scavenger which find applications in textile, plastic, paint, detergent, and rubber industry.
Solubility
Slightly soluble in water. Soluble in ethyl alcohol almost transparency.
Notes
Air and Moisture Sensitive. Store away from air and water/moisture. Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Store away from oxidizing agent.
RUO – Research Use Only
General References:
- B. J. Gregory; R. B. Moodie; and K. Schofield. Kinetics and mechanism of the Beckmann rearrangement of acetophenone oximes in sulphuric acid. J. Chem. Soc. B.,1970, 338-346.
- J Hes; LA Sternson. The metabolism of acetophenone oxime in rat liver homogenates. Drug Metabolism and Dispositon, 1974, 2 (4), 345-350.
- The dianion (n-BuLi) reacts to form heterocycles with electrophiles, e.g. methyl benzoate (or benzonitrile), CO2 or benzophenone: J. Org. Chem., 35, 1806 (1970); J. Chem. Soc. (C), 974 (1971); J. Heterocycl. Chem., 13, 449 (1976), respectively: