Search
Thermo Scientific Chemicals
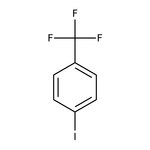
4-Iodobenzotrifluoride, 98%
CAS: 455-13-0 | C7H4F3I | 272.009 g/mol
化學識別
CAS455-13-0
IUPAC Name1-iodo-4-(trifluoromethyl)benzene
Molecular FormulaC7H4F3I
InChI KeySKGRFPGOGCHDPC-UHFFFAOYSA-N
SMILESFC(F)(F)C1=CC=C(I)C=C1
檢視更多
規格 規格表
規格表
Refractive Index1.5165-1.5205 @ 20?C
Appearance (Color)Clear colorless to orange
FormLiquid
Assay (GC)≥97.5%
4-Iodobenzotrifluoride may be employed as substrate with electron-deficient aromatic ring, during Mizoroki-Heck reaction with acrylic acid, to afford 4-trifluoromethylcinnnamic acid. It undergoes aminocarbonylation in DMF using phosphoryl chloride to give N,N-dimethyl-(4-trifluoromethyl)benzamide.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
4-Iodobenzotrifluoride may be employed as substrate with electron-deficient aromatic ring, during Mizoroki-Heck reaction with acrylic acid, to afford 4-trifluoromethylcinnnamic acid. It undergoes aminocarbonylation in DMF using phosphoryl chloride to give N,N-dimethyl-(4-trifluoromethyl)benzamide.
Solubility
Insoluble in water.
Notes
Light sensitive. Incompatible with strong oxidizing agents. Store in dark.
4-Iodobenzotrifluoride may be employed as substrate with electron-deficient aromatic ring, during Mizoroki-Heck reaction with acrylic acid, to afford 4-trifluoromethylcinnnamic acid. It undergoes aminocarbonylation in DMF using phosphoryl chloride to give N,N-dimethyl-(4-trifluoromethyl)benzamide.
Solubility
Insoluble in water.
Notes
Light sensitive. Incompatible with strong oxidizing agents. Store in dark.
RUO – Research Use Only
General References:
- Atsushi Ohtaka; Tomohiro Yamaguchi; Takuto Teratani; Osamu Shimomura; Ryôki Nomura. Linear polystyrene-stabilized PdO nanoparticle-catalyzed Mizoroki-Heck reactions in water. Molecules. 2011, 16 (11), 9067-9076.
- Kazushi Hosoi; Kyoko Nozaki; Tamejiro Hiyama. Carbon monoxide free aminocarbonylation of aryl and alkenyl iodides using DMF as an amide source. Organic Letters. 2002, 4 (17), 2849-2851.