Search
Thermo Scientific Chemicals
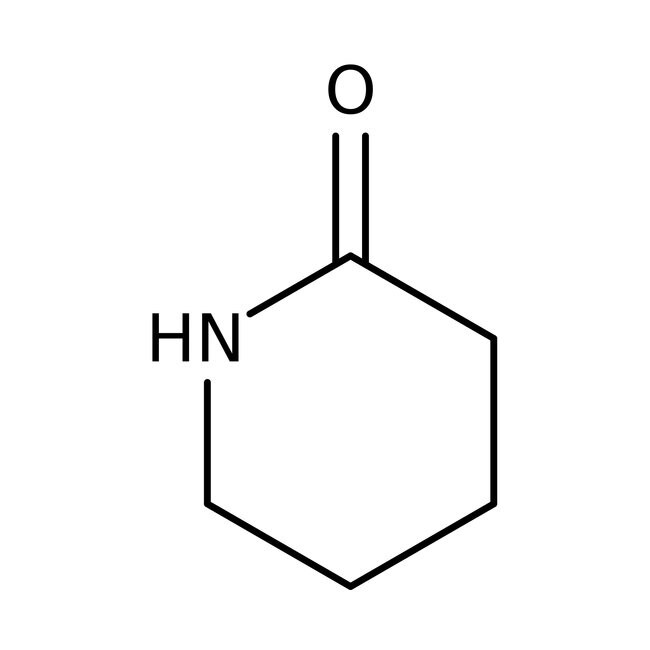
delta-Valerolactam, 98%
CAS: 675-20-7 | C5H9NO | 99.13 g/mol
化學識別
CAS675-20-7
IUPAC Namepiperidin-2-one
Molecular FormulaC5H9NO
InChI KeyXUWHAWMETYGRKB-UHFFFAOYSA-N
SMILESO=C1CCCCN1
檢視更多
規格 規格表
規格表
Appearance (Color)White to yellow or brown
FormCrystalline or fused solid
Assay (GC)≥97.5%
Identification (FTIR)Conforms
Water Content (Karl Fischer Titration)<2.0%
檢視更多
delta-Valerolactam is the starting material for a new route to hexahydrojulolidines, using intramolecular 1,4-cycloaddition. It is used as an intermediate in the preparation of other chemicals.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
delta-Valerolactam is the starting material for a new route to hexahydrojulolidines, using intramolecular 1,4-cycloaddition. It is used as an intermediate in the preparation of other chemicals.
Solubility
Soluble in water.
Notes
Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Keep away from strong oxidizing agents.
delta-Valerolactam is the starting material for a new route to hexahydrojulolidines, using intramolecular 1,4-cycloaddition. It is used as an intermediate in the preparation of other chemicals.
Solubility
Soluble in water.
Notes
Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Keep away from strong oxidizing agents.
RUO – Research Use Only
General References:
- Naoki Toyooka; Yasuko Yoshida; Yasuhito Yotsui and Takefumi Momose. 2-Piperidone Type of Chiral Building Block for 3-Piperidinol Alkaloid Synthesis. J. Org. Chem. 1999, 64 (13), 4914-4919.
- Tetsuji Kametani; Terumi Higa; Masuo Koizumi; Masataka Ihara; Keiichiro Fukumoto. Iminoketene cycloaddition. 2. Total syntheses of arborine, glycosminine, and rutecarpine by condensation of iminoketene with amides. J. Am. Chem. Soc. 1977, 99 (7), 2306-2309.
- Starting material for a new route to hexahydrojulolidines, using intramolecular 1,4-cycloaddition: J. Org. Chem., 58, 5041 (1993).
- For reaction as a heterodienophile in cycloaddition with Danishefsky's diene, see: Tris(6,6,7,7,8,8,8-heptafluoro-2,2-dimethyl-3,5-octanedionato) europium(III), 33541.