Search
Thermo Scientific Chemicals
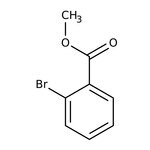
Methyl 2-bromobenzoate, 99%
CAS: 610-94-6 | C8H7BrO2 | 215.05 g/mol
化學識別
CAS610-94-6
IUPAC Namemethyl 2-bromobenzoate
Molecular FormulaC8H7BrO2
InChI KeySWGQITQOBPXVRC-UHFFFAOYSA-N
SMILESCOC(=O)C1=CC=CC=C1Br
檢視更多
規格 規格表
規格表
Appearance (Color)Clear colorless
Assay (GC)≥98.5%
Identification (FTIR)Conforms
Refractive Index1.5575-1.5605 @ 20?C
FormLiquid
Methyl 2-bromobenzoate under Heck reaction with acrylonitrile results in lactonization to give the benzofuran-2-one 3-(cyanomethyl)-2-coumaranone. It is an important raw material and intermediate used in Organic Synthesis, Pharmaceuticals, Agrochemicals
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Methyl 2-bromobenzoate under Heck reaction with acrylonitrile results in lactonization to give the benzofuran-2-one 3-(cyanomethyl)-2-coumaranone. It is an important raw material and intermediate used in Organic Synthesis, Pharmaceuticals, Agrochemicals
Solubility
Insoluble in water.
Notes
Store in a cool place. Keep away from oxidizing agents.
Methyl 2-bromobenzoate under Heck reaction with acrylonitrile results in lactonization to give the benzofuran-2-one 3-(cyanomethyl)-2-coumaranone. It is an important raw material and intermediate used in Organic Synthesis, Pharmaceuticals, Agrochemicals
Solubility
Insoluble in water.
Notes
Store in a cool place. Keep away from oxidizing agents.
RUO – Research Use Only
General References:
- Stephan Thorand.; Norbert Krause. Improved Procedures for the Palladium-Catalyzed Coupling of Terminal Alkynes with Aryl Bromides (Sonogashira Coupling). J. Org. Chem. 1998, 63 (23),8551-8553.
- Carsten Bolm.; Jens P. Hildebrand. Palladium-catalyzed carbonnitrogen bond formation: A novel, catalytic approach towards N-arylated sulfoximines. Tetrahedron Lett.. 1998, 39 (32),5731-5734.
- Heck reaction with acrylonitrile results in lactonization to give the benzofuran-2-one 3-(cyanomethyl)-2-coumaranone: J. Heterocycl. Chem., 32, 1587 (1995):