Search
Thermo Scientific Chemicals
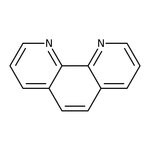
1,10-Phenanthroline, 99%, may contain up to 1.5% water
CAS: 66-71-7 | C12H8N2 | 180.21 g/mol
| 產品號碼 | Quantity |
|---|---|
| A13163.06 亦稱為 A13163-06 | 5 g |
化學識別
CAS66-71-7
IUPAC Name1,10-phenanthroline
Molecular FormulaC12H8N2
InChI KeyDGEZNRSVGBDHLK-UHFFFAOYSA-N
SMILESC1=CN=C2C(C=CC3=CC=CN=C23)=C1
檢視更多
規格 規格表
規格表
Assay (Non-aqueous acid-base Titration)≥98.5 to ≤101.5% (dry wt. basis)
Identification (FTIR)Conforms
Water Content (Karl Fischer Titration)≤1.5%
Melting Point (clear melt)113.0-121.0?C
Appearance (Color)White to cream
檢視更多
1,10-Phenanthroline, anhydrous is used as a chelating agent, used as bidendate nitrogen ligands in Heck type reactions as an alternative to phospine ligands. It is widely used as an indicator for alkyllithium reagents and an inhibitor of metallopeptidase enzymes. It is also used in the analysis formulations in end-user industries like food, beverage and pharmaceuticals.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
1,10-Phenanthroline is used as a chelating agent, used as bidendate nitrogen ligands in Heck type reactions as an alternative to phospine ligands. It is widely used as an indicator for alkyllithium reagents and an inhibitor of metallopeptidase enzymes. It is also used in the analysis formulations in end-user industries like food, beverage and pharmaceuticals.
Solubility
Soluble in acetone and ethanol. Slightly soluble in water.
Notes
Hygroscopic. Incompatible with strong oxidizing agents and strong acids. Store in a tightly closed container. Store in a cool, dry, well-ventilated area.
1,10-Phenanthroline is used as a chelating agent, used as bidendate nitrogen ligands in Heck type reactions as an alternative to phospine ligands. It is widely used as an indicator for alkyllithium reagents and an inhibitor of metallopeptidase enzymes. It is also used in the analysis formulations in end-user industries like food, beverage and pharmaceuticals.
Solubility
Soluble in acetone and ethanol. Slightly soluble in water.
Notes
Hygroscopic. Incompatible with strong oxidizing agents and strong acids. Store in a tightly closed container. Store in a cool, dry, well-ventilated area.
RUO – Research Use Only
General References:
- For use as bidentate nitrogen ligands in Heck type reactions as an alternative to phosphine ligands, see: Synlett., 11, 871 (1992).
- Indicator for titration of organolithium reagents, giving red charge-transfer complexes, and also in the titration of Grignard reagents: J. Organomet. Chem., 9, 165 (1967); Org. Synth. Coll., 9, 350, 653, 733 (1998).
- Reviews: 1,10-Phenanthroline: a versatile ligand: Chem. Soc. Rev., 23, 327 (1994); Coordination compounds as synthetic building blocks: Synlett, 1043 (2002).
- Petrov, E. G.; Marchenko, A.; Kapitanchuk, O. L.; Katsonis, N.; Fichou, D. Conductance Mechanism in a Linear Non-Conjugated Trimethylsilyl-Acetylene Molecule: Tunneling Through Localized States. Mol. Cryst. Liq. Cryst. 2014, 589 (1), 3-17.
- Wang, Y.; Zhang, Y.; Zhu, D.; Ma, K.; Ni, H.; Tang, G. Synthesis, structural characterization and theoretical approach of the tri(2-(2,6-dichlorophenyl)-1H-imidazo[4,5-f][1,10]phenanthroline) cobalt(II). Spectrochim. Acta, Part A 2015, 147 31-42.