Search
Thermo Scientific Chemicals
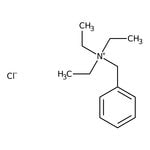
Benzyltriethylammonium chloride, 99%
CAS: 56-37-1 | C13H22ClN | 227.78 g/mol
化學識別
CAS56-37-1
規格 規格表
規格表
Appearance (Color)White to pale cream
FormCrystals or powder or crystalline powder
Assay (Titration ex Chloride)≥98.5% to ≤101.5% (Dry wt. basis) (UK Sourced Material)
Identification (FTIR)Conforms (UK Sourced Material)
Water Content (Karl Fischer Titration)≤2.5%
檢視更多
Benzyltriethylammonium chloride is used as a catalyst in the preparation of 2-phenylbutyronitrile from phenyl acetonitrile. It is involved in the Knoevenagel condensation of carbonyl compounds with active methylene compounds to give olefinic products. It acts as a phase transfer catalyst used in the alkylation reaction. It reacts with 1H-Pyridine-2-thione to get 2-Benzylsulfanyl-pyridine.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Benzyltriethylammonium chloride is used as a catalyst in the preparation of 2-phenylbutyronitrile from phenyl acetonitrile. It is involved in the Knoevenagel condensation of carbonyl compounds with active methylene compounds to give olefinic products. It acts as a phase transfer catalyst used in the alkylation reaction. It reacts with 1H-Pyridine-2-thione to get 2-Benzylsulfanyl-pyridine.
Solubility
Soluble in acetone, alcohol and water. Insoluble in ether.
Notes
Store in cool place. Hygroscopic. Incompatible with strong oxidizing agents.
Benzyltriethylammonium chloride is used as a catalyst in the preparation of 2-phenylbutyronitrile from phenyl acetonitrile. It is involved in the Knoevenagel condensation of carbonyl compounds with active methylene compounds to give olefinic products. It acts as a phase transfer catalyst used in the alkylation reaction. It reacts with 1H-Pyridine-2-thione to get 2-Benzylsulfanyl-pyridine.
Solubility
Soluble in acetone, alcohol and water. Insoluble in ether.
Notes
Store in cool place. Hygroscopic. Incompatible with strong oxidizing agents.
RUO – Research Use Only
General References:
- Phase-transfer catalyst (see Appendix 2) which has found wide application in the generation of carbenes from haloforms and related precursors: Org. Synth. Coll., 7, 12 (1990); 8, 223 (1993). Dihalocarbenes have been used to convert primary amines (t-butylamine) to isonitriles: Org. Synth. Coll., 6, 232 (1988), and in the dehydration of amides, thioamides and oximes to nitriles: Tetrahedron Lett., 2121 (1973). Ureas give cyanamides.
- For use in the formation of polysubstituted cyclopropanes by carbene-type addition to double bonds, see Ethyl chloroacetate, A15554.
- For use in the dialkylation of malonate esters to give cyclopropane derivatives, see: Synthesis, 54 (1985); Org. Synth. Coll., 7, 411 (1993).
- In combination with NaBr in acetonitrile, has been used for the mono-dealkylation of phosphonate diesters: Synthesis, 453 (1983).
- Ramos, M. D.; Gomez, G. I. G.; Gonzalez, N. S. Immobilization of Candida rugosa lipase on bentonite modified with benzyltriethylammonium chloride. J. Mol. Catal. B: Enzym. 2014, 99, 79-84.
- Wu, Y. C.; Leowanawat, P.; Sun, H. J.; Partridge, B. E.; Peterca, M.; Graf, R.; Spiess, H. W.; Zeng, X.; Ungar, G.; Hsu, C. S.; Heiney, P. A.; Percec, V. Complex Columnar Hexagonal Polymorphism in Supramolecular Assemblies of a Semifluorinated Electron-Accepting Naphthalene Bisimide. J. Am. Chem. Soc. 2015, 137 (2), 807-819.