Search
Thermo Scientific Chemicals
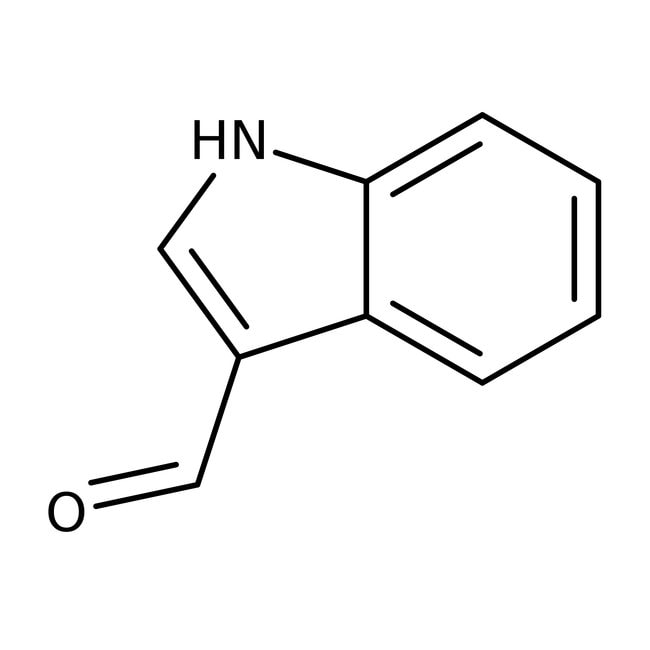
Indole-3-carboxaldehyde, 99%
CAS: 487-89-8 | C9H7NO | 145.161 g/mol
化學識別
CAS487-89-8
IUPAC Name1H-indole-3-carbaldehyde
Molecular FormulaC9H7NO
InChI KeyOLNJUISKUQQNIM-UHFFFAOYSA-N
SMILESO=CC1=CNC2=CC=CC=C12
檢視更多
規格 規格表
規格表
Appearance (Color)Pale yellow to yellow to pale brown or cream
FormCrystals or powder or crystalline powder
Assay (HPLC)≥98.5%
Free acid (titration)≤0.5%
Melting Point (clear melt)192.0-198.0?C
Indole-3-carboxaldehyde is a biochemical used to prepare analogs of the indole phytoalexin cyclobrassinin with NR1R2 group. It was also used as the starting material for the synthesis of higher order indoles including isoindolo[2,1-a]indoles, aplysinopsins, and 4-substituted-tetrahydrobenz[cd]indoles.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Indole-3-carboxaldehyde is a biochemical used to prepare analogs of the indole phytoalexin cyclobrassinin with NR1R2 group. It was also used as the starting material for the synthesis of higher order indoles including isoindolo[2,1-a]indoles, aplysinopsins, and 4-substituted-tetrahydrobenz[cd]indoles.
Solubility
Insoluble in water.
Notes
Air Sensitive. Avoid contact with air/oxygen. Incompatible with reducing agents.
Indole-3-carboxaldehyde is a biochemical used to prepare analogs of the indole phytoalexin cyclobrassinin with NR1R2 group. It was also used as the starting material for the synthesis of higher order indoles including isoindolo[2,1-a]indoles, aplysinopsins, and 4-substituted-tetrahydrobenz[cd]indoles.
Solubility
Insoluble in water.
Notes
Air Sensitive. Avoid contact with air/oxygen. Incompatible with reducing agents.
RUO – Research Use Only
General References:
- Deepa Sinha; Anjani K. Tiwari; Sweta Singh; Gauri Shukla; Pushpa Mishra; Harish Chandra; Anil K. Mishra. Synthesis, characterization and biological activity of Schiff base analogues of indole-3-carboxaldehyde.European Journal of Medicinal Chemistry.2008, 43 160-165.
- Robert E. Stutz. Enzymatic Formation of Indole-3-Carboxaldehyde from Indole-3-Acetic Acid.Plant Physiol.1958, 33 (3), 207-212..