Search
Thermo Scientific Chemicals
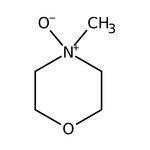
4-Methylmorpholine N-oxide monohydrate, 98+%
CAS: 70187-32-5 | C5H13NO3 | 135.163 g/mol
化學識別
CAS70187-32-5
IUPAC Name4-methylmorpholin-4-ium-4-olate
Molecular FormulaC5H11NO2
InChI KeyLFTLOKWAGJYHHR-UHFFFAOYSA-N
SMILESC[N+]1([O-])CCOCC1
檢視更多
規格 規格表
規格表
Assay (Non-aqueous acid-base Titration)≥98.0 to ≤102.0% (dry wt. basis)
Water Content (Karl Fischer Titration)10-16% (0.7-1.2 waters of hydration)
Appearance (Color)White to cream
FormCrystalline powder and/or Lumps
Solution TestClear colorless (ethanol, 5%)
4-Methylmorpholine N-oxide monohydrate is used as a solvent to prepare cellulose fibers. It is an oxidant and involved in the catalytic OsO4 oxidation of olefins to cis-1,2-diols. It is also involved in ruthenium catalyzed oxidation of alcohols to aldehydes and ketones.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
4-Methylmorpholine N-oxide monohydrate is used as a solvent to prepare cellulose fibers. It is an oxidant and involved in the catalytic OsO4 oxidation of olefins to cis-1,2-diols. It is also involved in ruthenium catalyzed oxidation of alcohols to aldehydes and ketones.
Solubility
Soluble in water.
Notes
Hygroscopic. Store in a cool place. Keep the container tightly closed. Incompatible with strong oxidizing agents.
4-Methylmorpholine N-oxide monohydrate is used as a solvent to prepare cellulose fibers. It is an oxidant and involved in the catalytic OsO4 oxidation of olefins to cis-1,2-diols. It is also involved in ruthenium catalyzed oxidation of alcohols to aldehydes and ketones.
Solubility
Soluble in water.
Notes
Hygroscopic. Store in a cool place. Keep the container tightly closed. Incompatible with strong oxidizing agents.
RUO – Research Use Only
General References:
- Stoichiometric oxidant permitting the use of Osmium(VIII) oxide, 12103, catalytically in the cis-hydroxylation of alkenes: Tetrahedron Lett., 1973 (1976): Org. Synth. Coll., 6, 342 (1988). For use in the asymmetric dihydroxylation (ADH) reaction, catalyzed by Osmium(VIII) oxide, 12103, see: J. Am. Chem. Soc., 110, 1968 (1988); Org. Synth. Coll., 9, 383 (1998).
- In the presence of Ru catalysts such as RuCl3 or Dichlorotris(triphenyl phosphine) ruthenium(II) , L00373, oxidizes various types of alcohol to carbonyl compounds: Tetrahedron Lett., 2503 (1976). See also Tetra-n-propyl ammonium perruthenate(VII) , B24511.
- In the presence of 4A molecular sieves in acetonitrile, activated (e.g. benzylic) halides can be oxidized to aldehydes and ketones: Synth. Commun., 22, 1967 (1992).
- Boronic esters formed by treatment of aryl bromides in THF with n-BuLi, followed by trimethyl borate, can be oxidized in situ to phenols without isolation of the boronic acid: Synlett, 931 (1995).
- Chapelat, J.; Hengartner, U.; Chougnet, A.; Liu, K.; Huebbe, P.; Rimbach, G.; Woggon, W. D. Asymmetric Synthesis and Biological Activity of nor-α-Tocopherol, a New Vitamin E Analogue. ChemBioChem 2011, 12 (1), 118-124.
- Tong, L. H.; Guenee, L.; Williams, A. F. Pentasubstituted Ferrocene and Dirhodium(II) Tetracarboxylate as Building Blocks for Discrete Fullerene-Like and Extended Supramolecular Structures. Inorg. Chem. 2011, 50 (6), 2450-2457.