Search
Thermo Scientific Chemicals
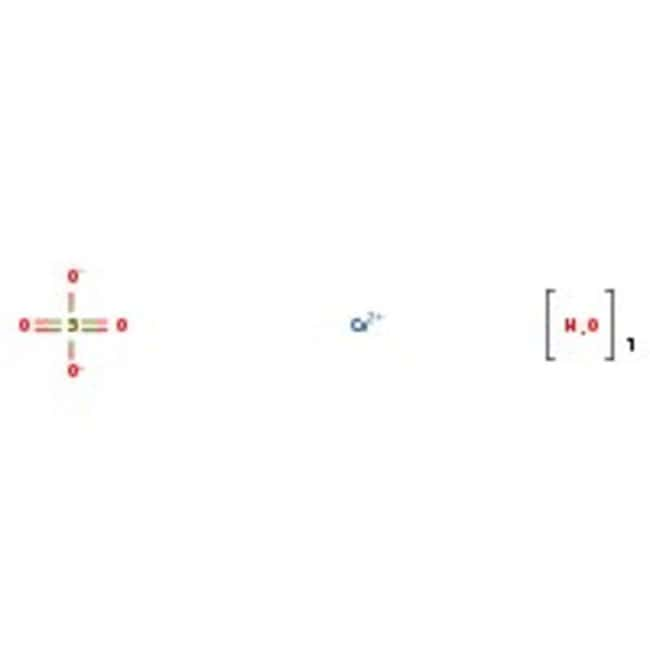
Cobalt(II) sulfate heptahydrate, 98%
CAS: 10026-24-1 | CoH14O11S | 281.09 g/mol
化學識別
CAS10026-24-1
IUPAC Nameλ²-cobalt(2+) heptahydrate sulfate
Molecular FormulaCoH14O11S
InChI KeyMEYVLGVRTYSQHI-UHFFFAOYSA-L
SMILESO.O.O.O.O.O.O.[Co++].[O-]S([O-])(=O)=O
檢視更多
規格 規格表
規格表
Appearance (Color)Red to dark red or red-brown
Elemental AnalysisCobalt (Co) : 20.12-21.49% (as heptahydrate)
FormCrystals or crystalline powder
Assay from Supplier's CofA≥97.5 to ≤102.5% (based on Cobalt EA)
Cobalt(II) sulfate heptahydrate is used in the preparation of pigments, as well as in the manufacture of other cobalt salts, which is used in porcelains and glass. It is used in storage batteries and electroplating baths, sympathetic inks, and as an additive to soils and animal feeds.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Cobalt(II) sulfate heptahydrate is used in the preparation of pigments, as well as in the manufacture of other cobalt salts, which is used in porcelains and glass. It is used in storage batteries and electroplating baths, sympathetic inks, and as an additive to soils and animal feeds.
Solubility
Soluble in water.
Notes
Incompatible with strong oxidizing agents.
Cobalt(II) sulfate heptahydrate is used in the preparation of pigments, as well as in the manufacture of other cobalt salts, which is used in porcelains and glass. It is used in storage batteries and electroplating baths, sympathetic inks, and as an additive to soils and animal feeds.
Solubility
Soluble in water.
Notes
Incompatible with strong oxidizing agents.
RUO – Research Use Only
General References:
- Tang, C.; Wei, X.; Jiang, Y.; Wu, X.; Han, L.; Wang, K.; Chen, J. Cobalt-Doped MnO2 Hierarchical Yolk-Shell Spheres with Improved Supercapacitive Performance. J. Phys. Chem. C 2015, 119 (16), 8465-8471.
- Mayra, Q.; Kim, W. Agglomeration of Ni-Rich Hydroxide in Reaction Crystallization: Effect of Taylor Vortex Dimension and Intensity. Crys. Growth Des. 2015, 15 (4), 1726-1734.