Search
Thermo Scientific Chemicals
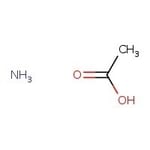
Ammonium acetate, 97%
CAS: 631-61-8 | C2H7NO2 | 77.083 g/mol
化學識別
CAS631-61-8
IUPAC Nameammonium acetate
Molecular FormulaC2H7NO2
InChI KeyUSFZMSVCRYTOJT-UHFFFAOYSA-N
SMILES[NH4+].CC([O-])=O
檢視更多
規格 規格表
規格表
FormCrystals or powder or crystalline powder
Identification (FTIR)Conforms
Assay from Supplier's CofAAmmonia as ammonium acetate: ≥96.0% to ≤104.0%
Water Content (Karl Fischer Titration)≤5.0%
Appearance (Color)White
Ammonium acetate is widely utilized as a catalyst in the Knoevenagel condensation. It is the primary source of ammonia in the Borch reaction in organic synthesis. It is used with distilled water to make a protein precipitating reagent. It acts as a buffer for electrospray ionization (ESI) mass spectrometry of proteins and other molecules and as mobile phases for high performance liquid chromatography (HPLC). Sometimes, it is used as a biodegradable de-icing agent and an acidity regulator in food additives.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Ammonium acetate is widely utilized as a catalyst in the Knoevenagel condensation. It is the primary source of ammonia in the Borch reaction in organic synthesis. It is used with distilled water to make a protein precipitating reagent. It acts as a buffer for electrospray ionization (ESI) mass spectrometry of proteins and other molecules and as mobile phases for high performance liquid chromatography (HPLC). Sometimes, it is used as a biodegradable de-icing agent and an acidity regulator in food additives.
Solubility
Soluble in water, methanol, acetone and liquid ammonia.
Notes
Hygroscopic. Incompatible with strong oxidizing agents and strong acids.
Ammonium acetate is widely utilized as a catalyst in the Knoevenagel condensation. It is the primary source of ammonia in the Borch reaction in organic synthesis. It is used with distilled water to make a protein precipitating reagent. It acts as a buffer for electrospray ionization (ESI) mass spectrometry of proteins and other molecules and as mobile phases for high performance liquid chromatography (HPLC). Sometimes, it is used as a biodegradable de-icing agent and an acidity regulator in food additives.
Solubility
Soluble in water, methanol, acetone and liquid ammonia.
Notes
Hygroscopic. Incompatible with strong oxidizing agents and strong acids.
RUO – Research Use Only
General References:
- Xia, S.; Zhang, H. Density Functional Theory Study of Selective Deacylation of Aromatic Acetate in the Presence of Aliphatic Acetate under Ammonium Acetate Mediated Conditions. J. Org. Chem. 2014, 79 (13), 6135-6142.
- Zhang, J.; Yao, J.; Liu, J.; Xue, S.; Li, Y.; Wang, C. Four-component reaction between naphthols, substituted β-nitrostyrenes, substituted benzaldehydes and ammonium acetate in water-PEG-400: an approach to construct polysubstituted naphthofuranamines. RSC Adv. 2015, 5 (60), 48580-48585.

.png-150.jpg)

