Search
Thermo Scientific Chemicals
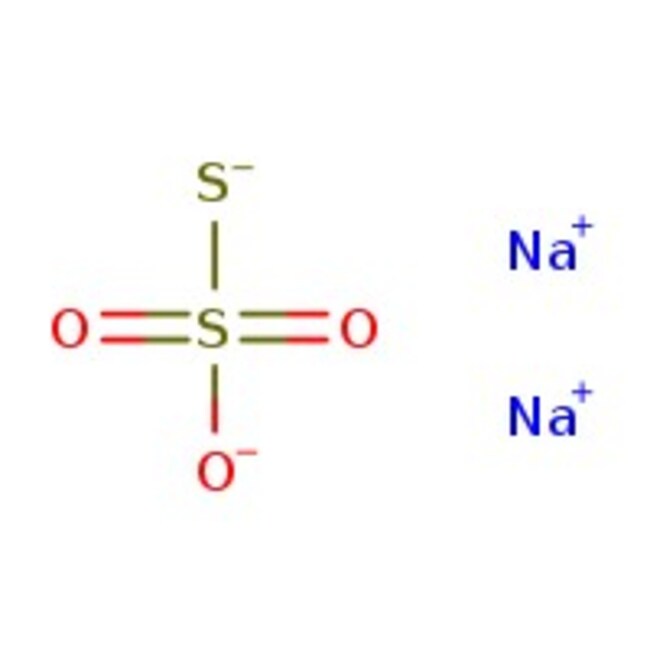
Sodium thiosulfate, anhydrous, 98+%
CAS: 7772-98-7 | Na2O3S2 | 158.10 g/mol
化學識別
CAS7772-98-7
IUPAC Namedisodium sulfanidesulfonate
Molecular FormulaNa2O3S2
InChI KeyAKHNMLFCWUSKQB-UHFFFAOYSA-L
SMILES[Na+].[Na+].[O-]S([S-])(=O)=O
檢視更多
規格 規格表
規格表
Identification (FTIR)Conforms
Assay (Iodometric Titration)≥98.0% (on dried substance)
Loss on Drying≤2.0% (2hr, 105?C)
Appearance (Color)White
FormCrystals or powder or crystalline powder
Sodium thiosulfate is used in volumetric analysis to estimate the concentration of certain compounds in solution (hydrogen peroxide) as well as chlorine content in bleaching powder and water. It is used to prepare S-alkylthiosulfonates. Furthermore, it is used in the tanning of leather and in photographic processing. It is used as an antidote to cyanide poisoning and acts as a safe disposal medium in chemistry laboratories when working with bromine, iodine and other strong oxidizers. It is an important component in hand warmers and heating pads.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Sodium thiosulfate is used in volumetric analysis to estimate the concentration of certain compounds in solution (hydrogen peroxide) as well as chlorine content in bleaching powder and water. It is used to prepare S-alkylthiosulfonates. Furthermore, it is used in the tanning of leather and in photographic processing. It is used as an antidote to cyanide poisoning and acts as a safe disposal medium in chemistry laboratories when working with bromine, iodine and other strong oxidizers. It is an important component in hand warmers and heating pads.
Solubility
Soluble in water. Insoluble in alcohol.
Notes
Hygroscopic. Incompatible with strong acids, strong oxidizing agents, iodine and mercury.
Sodium thiosulfate is used in volumetric analysis to estimate the concentration of certain compounds in solution (hydrogen peroxide) as well as chlorine content in bleaching powder and water. It is used to prepare S-alkylthiosulfonates. Furthermore, it is used in the tanning of leather and in photographic processing. It is used as an antidote to cyanide poisoning and acts as a safe disposal medium in chemistry laboratories when working with bromine, iodine and other strong oxidizers. It is an important component in hand warmers and heating pads.
Solubility
Soluble in water. Insoluble in alcohol.
Notes
Hygroscopic. Incompatible with strong acids, strong oxidizing agents, iodine and mercury.
RUO – Research Use Only
General References:
- Wendroth, S. M.; Heady, T. N.; Haverstick, D. M.; Bachmann, L. M.; Scott, M. G.; Boyd, J. C.; Bruns, D. E. Falsely increased chloride and missed anion gap elevation during treatment with sodium thiosulfate. Clin. Chim. Acta 2014, 431, 77-79.
- Chen, N. X.; O’Neill, K.; Akl, N. K.; Moe, S. M. Adipocyte induced arterial calcification is prevented with sodium thiosulfate. Biochem. Biophys. Res. Commun. 2014, 449 (1), 151-156.