Search
Thermo Scientific Chemicals
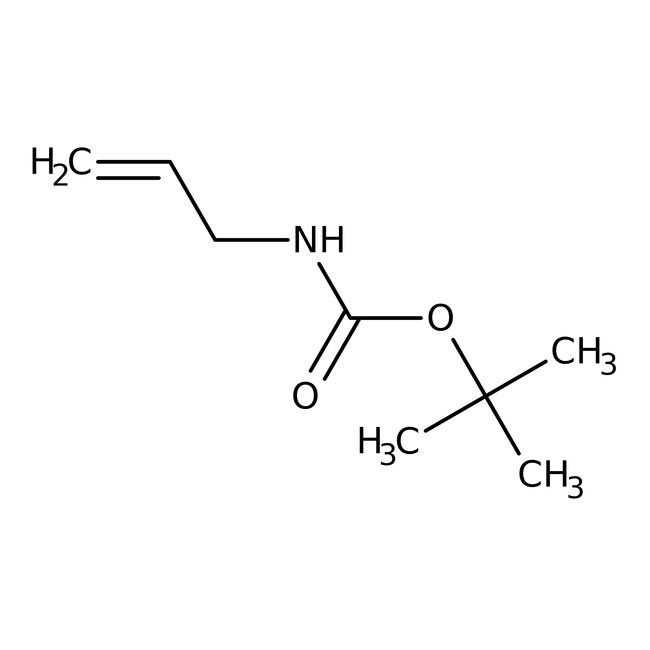
tert-Butyl N-allylcarbamate, 97%
CAS: 78888-18-3 | C8H15NO2 | 157.213 g/mol
化學識別
CAS78888-18-3
IUPAC Nametert-butyl N-(prop-2-en-1-yl)carbamate
Molecular FormulaC8H15NO2
InChI KeyAWARHXCROCWEAK-UHFFFAOYSA-N
SMILESCC(C)(C)OC(=O)NCC=C
檢視更多
規格 規格表
規格表
Appearance (Color)White to cream to pale yellow
FormCrystals or powder or crystalline powder or fused/lumpy solid
Assay (GC)≥96.0%
tert-Butyl N-allylcarbamate is used in the synthesis of isoxazolidines. Synthesis of 5-substituted thiazolidin-2-ones is from the reaction of xanthates and tert-butyl N-allylcarbamates .
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
tert-Butyl N-allylcarbamate is used in the synthesis of isoxazolidines. Synthesis of 5-substituted thiazolidin-2-ones is from the reaction of xanthates and tert-butyl N-allylcarbamates .
Solubility
Insoluble in water. Soluble in methanol.
Notes
Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Store away from strong oxidizing agents.
tert-Butyl N-allylcarbamate is used in the synthesis of isoxazolidines. Synthesis of 5-substituted thiazolidin-2-ones is from the reaction of xanthates and tert-butyl N-allylcarbamates .
Solubility
Insoluble in water. Soluble in methanol.
Notes
Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Store away from strong oxidizing agents.
RUO – Research Use Only
General References:
- Antonio Rescifinaa; Ugo Chiacchioa; Antonino Corsaroa; Anna Pipernob; Roberto Romeob. Isoxazolidinyl polycyclic aromatic hydrocarbons as DNA-intercalating antitumor agents. European Journal of Medicinal Chemistry. 2011, 46 (1) ,129-136.
- Zhongyan Huang,; Jiaxi Xu. Efficient synthesis of 5-substituted thiazolidin-2-ones from xanthates and tert-butyl N-allylcarbamates . Tetrahedron. 2013, 69 (48), 10272-10278.