Search
Thermo Scientific Chemicals
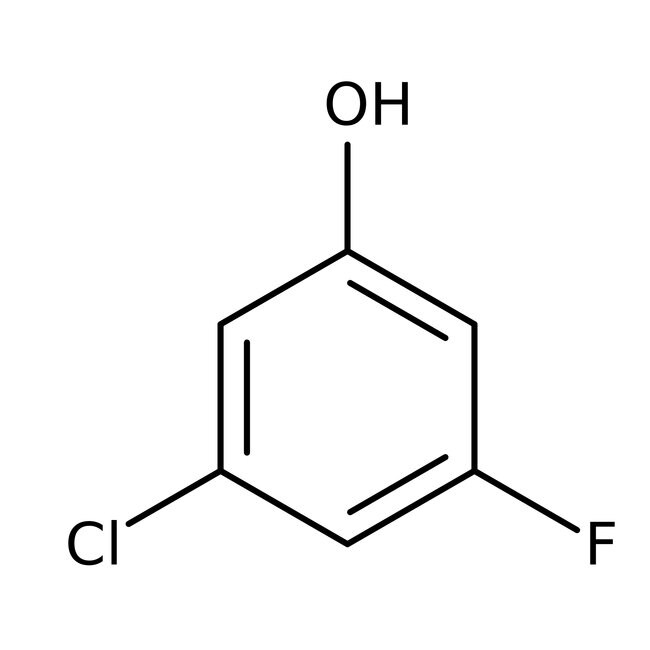
3-Chloro-5-fluorophenol, 98%
CAS: 202982-70-5 | C6H4ClFO | 146.545 g/mol
化學識別
CAS202982-70-5
IUPAC Name3-chloro-5-fluorophenol
Molecular FormulaC6H4ClFO
InChI KeyZMFBYTAAMHWQHD-UHFFFAOYSA-N
SMILESOC1=CC(F)=CC(Cl)=C1
檢視更多
規格 規格表
規格表
Refractive Index1.5345-1.5385 @ 20?C
Assay (GC)≥97.5%
Appearance (Color)Clear colorless to pale yellow
FormLiquid
3-Chloro-5-fluorophenol is used as an intermediate in organic synthesis. It plays an important role in the preparation of substituted amides as antagonists and/or inverse agonists of cannabinoid-1 receptor, substituted N-(arylmethyl)aryloxy arylcarboxamide and heteroarylcarboxamide antagonists for the PGE2 receptor EP4 for the treatment of pain, inflammation, osteoarthritis, and rheumatoid arthritis.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
3-Chloro-5-fluorophenol is used as an intermediate in organic synthesis. It plays an important role in the preparation of substituted amides as antagonists and/or inverse agonists of cannabinoid-1 receptor, substituted N-(arylmethyl)aryloxy arylcarboxamide and heteroarylcarboxamide antagonists for the PGE2 receptor EP4 for the treatment of pain, inflammation, osteoarthritis, and rheumatoid arthritis.
Solubility
Immiscible with water.
Notes
Incompatible with strong oxidizing agents.
3-Chloro-5-fluorophenol is used as an intermediate in organic synthesis. It plays an important role in the preparation of substituted amides as antagonists and/or inverse agonists of cannabinoid-1 receptor, substituted N-(arylmethyl)aryloxy arylcarboxamide and heteroarylcarboxamide antagonists for the PGE2 receptor EP4 for the treatment of pain, inflammation, osteoarthritis, and rheumatoid arthritis.
Solubility
Immiscible with water.
Notes
Incompatible with strong oxidizing agents.
RUO – Research Use Only
Huang, H.; Ji, H.; Li, H.; Jing, Q.; Labby, K. J.; Martasek, P.; Roman, L. J.; Poulos, T. L.; Silverman, R. B. Selective Monocationic Inhibitors of Neuronal Nitric Oxide Synthase. Binding Mode Insights from Molecular Dynamics Simulations. J. Am. Chem. Soc. 2012, 134 (28), 11559-11572.