Search
Thermo Scientific Chemicals
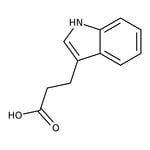
Indole-3-propionic acid, 98%
CAS: 830-96-6 | C11H11NO2 | 189.21 g/mol
化學識別
CAS830-96-6
規格 規格表
規格表
Appearance (Color)Pale cream to cream or pale yellow or pale brown
FormPowder
Assay from Supplier's CofA≥97.5%
Identification (FTIR)Conforms
Indole-3-propionic acid is a reactant for preparation of fluorescent analogues of strigolactones, melanocortin receptors ligands, histamine H4 receptor agonists, and NR2B/NMDA receptor antagonists. It is also used as an important raw material and intermediate used in organic Synthesis, pharmaceuticals, agrochemicals and dyestuff. It is also used to Study as an adjunct to improve perfusion after liver transplant.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Indole-3-propionic acid is a reactant for preparation of fluorescent analogues of strigolactones, melanocortin receptors ligands, histamine H4 receptor agonists, and NR2B/NMDA receptor antagonists. It is also used as an important raw material and intermediate used in organic Synthesis, pharmaceuticals, agrochemicals and dyestuff. It is also used to Study as an adjunct to improve perfusion after liver transplant.
Solubility
It is slightly soluble in water.
Notes
Keep container tightly closed in a dry and well-ventilated place. Keep away from oxidizing agents.
Indole-3-propionic acid is a reactant for preparation of fluorescent analogues of strigolactones, melanocortin receptors ligands, histamine H4 receptor agonists, and NR2B/NMDA receptor antagonists. It is also used as an important raw material and intermediate used in organic Synthesis, pharmaceuticals, agrochemicals and dyestuff. It is also used to Study as an adjunct to improve perfusion after liver transplant.
Solubility
It is slightly soluble in water.
Notes
Keep container tightly closed in a dry and well-ventilated place. Keep away from oxidizing agents.
RUO – Research Use Only
General References:
- Yuan-Hsiao Su et. al. Solution-phase parallel synthesis and screening of anti-tumor activities from fenbufen and ethacrynic acid libraries. Bioorganic & Medicinal Chemistry Letters.2011, 21,(5), 1320-1324 .
- Francis Giraud et. al. Synthesis ofN-aryl-3-(indol-3-yl)propanamides and their immunosuppressive activities. Bioorganic & Medicinal Chemistry Letters.2011, 20,(17), 5203-5206.