Search
Thermo Scientific Chemicals
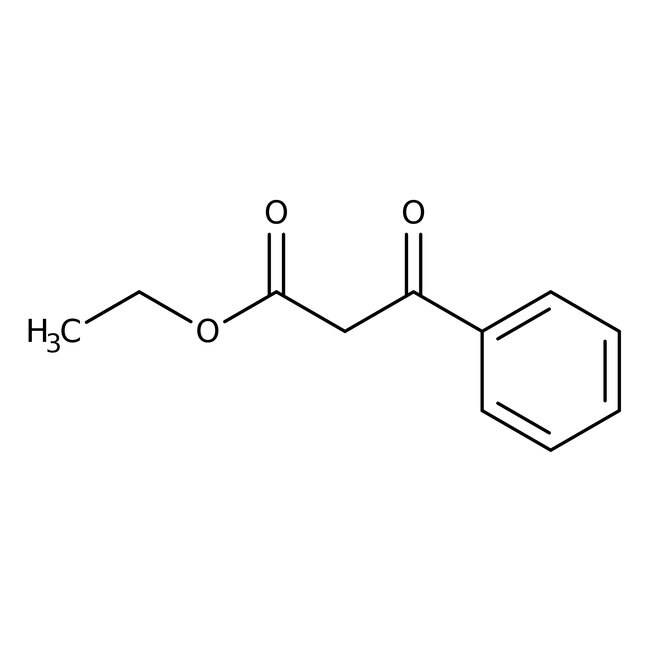
Ethyl benzoylacetate, 90+%
CAS: 94-02-0 | C11H12O3 | 192.214 g/mol
化學識別
CAS94-02-0
IUPAC Nameethyl 3-oxo-3-phenylpropanoate
Molecular FormulaC11H12O3
InChI KeyGKKZMYDNDDMXSE-UHFFFAOYSA-N
SMILESCCOC(=O)CC(=O)C1=CC=CC=C1
檢視更多
規格 規格表
規格表
Assay (HPLC)≥90.0%
Appearance (Color)Clear colorless to white to yellow to orange to brown
Refractive Index1.4875-1.5675 @ 20?C
FormLiquid
Ethyl benzoylacetate is used as a flavoring agent and an intermediate in organic synthesis. Further, it is used in the preparation of triazipinones, iodonium ylides, ethyl 2-fluoro-2-benzolyacetate and benzo[c]furan-4,7-diones. It is involved in the Clisen condensation reaction with malononitrile to prepare 2-cyano-5-phenyl-3,5-dioxopentanonitrile.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Ethyl benzoylacetate is used as a flavoring agent and an intermediate in organic synthesis. Further, it is used in the preparation of triazipinones, iodonium ylides, ethyl 2-fluoro-2-benzolyacetate and benzo[c]furan-4,7-diones. It is involved in the Clisen condensation reaction with malononitrile to prepare 2-cyano-5-phenyl-3,5-dioxopentanonitrile.
Solubility
Miscible with ethanol and ether. Immiscible with water.
Notes
Air and light sensitive. Incompatible with acids, bases, oxidizing agents and reducing agents.
Ethyl benzoylacetate is used as a flavoring agent and an intermediate in organic synthesis. Further, it is used in the preparation of triazipinones, iodonium ylides, ethyl 2-fluoro-2-benzolyacetate and benzo[c]furan-4,7-diones. It is involved in the Clisen condensation reaction with malononitrile to prepare 2-cyano-5-phenyl-3,5-dioxopentanonitrile.
Solubility
Miscible with ethanol and ether. Immiscible with water.
Notes
Air and light sensitive. Incompatible with acids, bases, oxidizing agents and reducing agents.
RUO – Research Use Only
General References:
- Has been used in a one-pot synthesis of ɑ-substituted acrylic esters: Synth. Commun., 18, 1213 (1988):
- Compare also Ethyl acetoacetate, A12544.
- Rajesh, N.; Prajapati, D. Indium-catalyzed, novel route to beta,beta-disubstituted indanones via tandem Nakamura addition-hydroarylation-decarboxylation sequence. Chem. Commun. 2015, 51 (16), 3347-3350.
- Ni, Q.; Xiong, J.; Song, X.; Raabe, G.; Enders, D. NHC-catalyzed activation of alpha,beta-unsaturated N-acyltriazoles: an easy access to dihydropyranones. Chem. Commun. 2015, 51 (78), 14628-14631.