Search
Thermo Scientific Chemicals
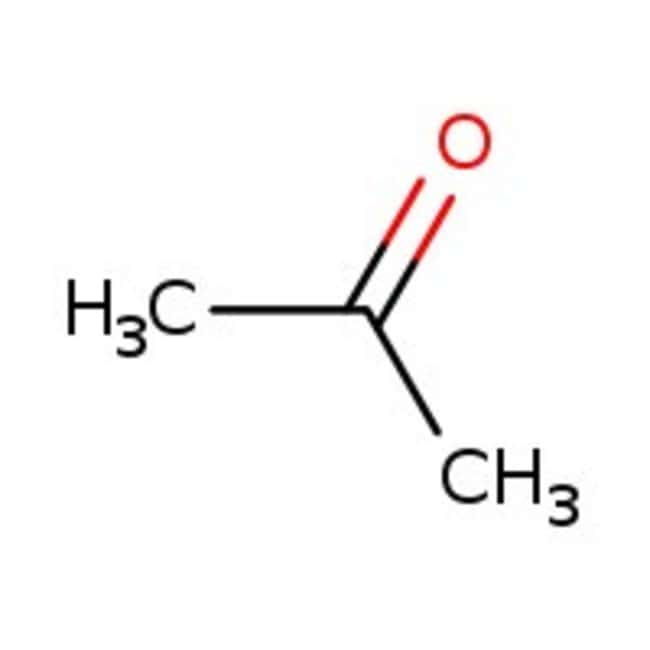
Acetone, 99+%
CAS: 67-64-1 | C3H6O | 58.08 g/mol
化學識別
CAS67-64-1
IUPAC Namepropan-2-one
Molecular FormulaC3H6O
InChI KeyCSCPPACGZOOCGX-UHFFFAOYSA-N
SMILESCC(C)=O
檢視更多
規格 規格表
規格表
Appearance (Color)Clear colorless
FormLiquid
Assay (GC)≥99.0% (non-U.S. specification)
Assay from Suppliers CofA≥99.0% (U.S. specification)
CommentSpecification differs for U.S. and non-U.S. material where indicated
檢視更多
Acetone is a polar, aprotic solvent used in the synthesis and isolation of both organic and inorganic compounds and complexes. It plays a vital role in Jones oxidation. It is utilized for rinsing laboratory glasswares due to its low cost and volatility. It is used in the manufacture of various chemicals such as methyl isobutyl ketone, mesityl oxide, diacetone alcohol, acetic acid, iodoform, bromoform, rayon, photographic films, paint and varnish removers. It is also useful in the synthesis of t-butanesulfinamide, terpenes, thiol-stabilized gold colloids, calixphyrin macrocycles and trispyrazolylborate platinum compounds. It plays an important role in protein crystallization.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Acetone is a polar, aprotic solvent used in the synthesis and isolation of both organic and inorganic compounds and complexes. It plays a vital role in Jones oxidation. It is utilized for rinsing laboratory glasswares due to its low cost and volatility. It is used in the manufacture of various chemicals such as methyl isobutyl ketone, mesityl oxide, diacetone alcohol, acetic acid, iodoform, bromoform, rayon, photographic films, paint and varnish removers. It is also useful in the synthesis of t-butanesulfinamide, terpenes, thiol-stabilized gold colloids, calixphyrin macrocycles and trispyrazolylborate platinum compounds. It plays an important role in protein crystallization.
Solubility
Miscible with water, benzene, alcohol, dimethylformamide, ether, chloroform and most oils.
Notes
Incompatible with bases, oxidizing agents and reducing agents.
Acetone is a polar, aprotic solvent used in the synthesis and isolation of both organic and inorganic compounds and complexes. It plays a vital role in Jones oxidation. It is utilized for rinsing laboratory glasswares due to its low cost and volatility. It is used in the manufacture of various chemicals such as methyl isobutyl ketone, mesityl oxide, diacetone alcohol, acetic acid, iodoform, bromoform, rayon, photographic films, paint and varnish removers. It is also useful in the synthesis of t-butanesulfinamide, terpenes, thiol-stabilized gold colloids, calixphyrin macrocycles and trispyrazolylborate platinum compounds. It plays an important role in protein crystallization.
Solubility
Miscible with water, benzene, alcohol, dimethylformamide, ether, chloroform and most oils.
Notes
Incompatible with bases, oxidizing agents and reducing agents.
RUO – Research Use Only
General References:
- Precursor of dimethyldioxirane, by reaction with K monopersulfate, and isolation in solution by low temperature distillation. For details, see: Chem. Ber., 124, 2377 (1991). It is an excellent reagent for rapid, stereospecific epoxidation of alkenes under mild conditions: J. Org. Chem., 50, 2847 (1985); Org. Synth. Coll., 9, 288 (1998). For improved work-up procedure allowing the possibility of more concentrated solutions or the isolation of acetone-free material, see: Tetrahedron Lett., 37, 3585 (1996). For biphasic, environmentally-friendly system (EtOAc-H2 O), see: Org. Process Res. Dev., 6, 407 (2002). Selective oxidation of phosphorus ylides occurs under mild conditions, providing a route to vicinal tricarbonyl derivatives: J. Org. Chem., 60, 8231 (1995). For conversion of ɑ-bromo-ß-dicarbonyls to vicinal tricarbonyls, see: Tetrahedron Lett., 36, 7735 (1995). For formation of singlet oxygen by deoxygenation of heteroaromatic N-oxides, e.g. 4-diethylaminopyridine N-oxide, see: J. Chem. Soc., Chem. Commun., 1831 (1995). For reviews of dioxiranes, see: Acc. Chem. Res., 22, 205 (1989); Chem. Rev., 89, 1187 (1989).
- Navarro, R. M.; Guil-Lopez, R.; Ismail, A. A.; Al-Sayari, S. A.; Fierro, J. L. G. Ni-and PtNi-catalysts supported on Al2O3 for acetone steam reforming: Effect of the modification of support with Ce, La and Mg. Catal. Today 2015, 242, 60-70.
- Quesada, J.; Faba, L.; Díaz, E.; Bennici, S.; Auroux, A.; Ordóñez, S. Role of surface intermediates in the deactivation of Mg Zr mixed oxides in acetone self-condensation: A combined DRIFT and ex situ characterization approach. J. Catal. 2015, 329, 1-9.