Search
GlobalFiler STR Kits

GlobalFiler STR kits offer superior discriminatory power for international data sharing
Around the world, forensic DNA labs are being asked to do more with less. This is why the Applied Biosystems GlobalFiler IQC, GlobalFiler, and GlobalFiler Express PCR amplification kits combine reduced amplification time with exceptional discrimination power, even on the most challenging sample types.
As global forensic DNA databases rapidly expand, so does the need for more discriminating STR multiplexes that can maximize loci overlap. That’s why GlobalFiler kits incorporate the most commonly used loci—all in a single multiplex, 6-dye configuration kit. GlobalFiler kits contain all markers recommended for inclusion by the CODIS Core Loci Working Group and those markers commonly used in Europe. The multiplex selection of markers reduces the risk of adventitious matches while enabling more effective cross-border data sharing. And the kits are backed by training, service, and support from Thermo Fisher Scientific.
Key benefits
The GlobalFiler kit is also available with an Internal Quality Control system, or IQC, as part of a fully integrated and validated forensic workflow. The IQC system consists of two synthetic sequences with specific primers for each of the targets (IQCS and IQCL) and provides positive confirmation of sample amplification, but also indicates adverse conditions with compromised amplification, such as the presence of PCR inhibitors. The IQC system provides additional confidence in genotyping results and can help forensic scientists distinguish, for example, between inhibited and degraded DNA samples.

GlobalFiler kits are manufactured at our location in Warrington, UK, at a facility that meets the guidelines for ISO 18385 certification. We have made significant investments across all aspects of production to minimize human DNA contamination. The result: powerful forensic DNA-grade solutions that enable you to provide answers with certainty and confidence.

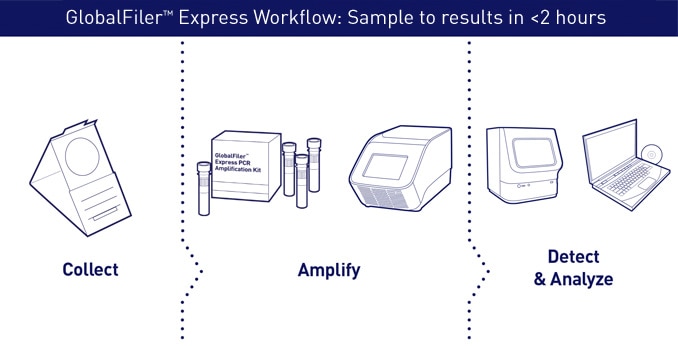
Turn samples into answers in less than 2 hours
This breakthrough chemistry enables up to 5 times faster amplification time than previous-generation kits:
- GlobalFiler Express Kit for single source samples: ~40 minute amplification
- GlobalFiler Kit for casework samples: ~80 minute amplification

Typical workflow for up to 48 samples using Applied Biosystems direct PCR amplification kits, genetic analyzers and expert system analysis software.
Up to 9 orders of magnitude more discrimination power
- 24 loci multiplex enables up to 9 orders of magnitude more discrimination power than previous-generation kits
- Includes 10 powerful mini-STR loci for increased information recovery from heavily degraded samples
- Enhanced buffer system enables superior performance on inhibited samples
- Improved sensitivity and flexibility to add up to 15 μL of sample input volume enables increased allele recovery from low level samples

The GlobalFiler Kit multiplex configuration includes all 24 loci with only 1 locus partially exceeding 400 base pairs. 10 mini-STR loci lie completely below 220 base pairs for enhanced performance on degraded samples, and all gender-specific markers are located in the green VIC channel for convenience of interpretation. The IQCS and IQCL markers are only present in GlobalFiler IQC kit.

The GlobalFiler Kit allelic ladder includes 343 alleles featuring expanded marker ranges for many loci while maintaining interlocus spacing. The expanded panel and virtual bins (589 total alleles) minimize OL allele calls and facilitate more accurate and efficient automated genotyping.
As global forensic DNA databases rapidly expand, so does the need for more discriminating STR multiplexes that can maximize loci overlap. That’s why GlobalFiler kits incorporate as many commonly used loci as possible—all in a single multiplex.
- GlobalFiler kits contain all markers recommended for inclusion by the CODIS Core Loci Working Group, including all markers commonly used in Europe
- Reduces the risk of adventitious matches while enabling more effective cross-border data sharing
- The GlobalFiler, GlobalFiler IQC, and GlobalFiler Express kits are approved by the FBI’s National DNA Index System (NDIS) Board for use by laboratories generating offender DNA profiles for inclusion in the U.S. National NDIS database

“Improving quality control processes and performance checks is a continuous challenge in forensics, more particularly for rapid, high-throughput, and reliable generation of DNA profiles from casework samples. The Internal Quality Control (IQC) system combined into GlobalFiler kits generates a powerful and controlled STR multiplex PCR assay for better and faster decision making and data interpretation.”
—Dr Sylvain Hubac
Research and innovation manager
High throughput DNA unit for casework samples
Forensic Science laboratory of the French Gendarmerie, France

Pablo Martín, Lourdes Fernández de Simón, Gracia Luque, María José Farfán, Antonio Alonso
DOI: http://www.fsigenetics.com/article/S1872-4973(14)00138-0/fulltext
p68–78
Published online: July 15, 2014
For Forensic or Paternity Use Only.
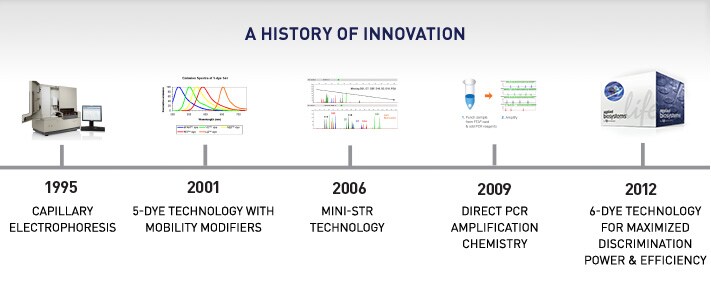
For the past 16 years, Thermo Fisher Scientific has led the way in the evolution of STR technology. And today, GlobalFiler kits utilize breakthrough 6-dye chemistry to enable optimal performance and unprecedented discrimination.
- Minimizes the number of loci that are >400 bp, which decreases drop-outs with degraded and challenging samples
- Increases the number of miniSTRs—the GlobalFiler Kits include 10 STRs completely under 220 bp
- Allows for better spacing between adjacent markers and expansion of marker ranges to minimize off-ladder allele calls


Using a 6-dye configuration enables optimal performance, efficiency, data recovery and genotyping accuracy.
Key features of the GlobalFiler kits
| GlobalFiler kit | GlobalFiler IQC kit | GlobalFiler Express kit | |
| High discrimination power | 24-marker multiplex assay including 3 gender markers and the highly discriminatory SE33 locus | ||
| Number of dyes | 6 | ||
| Mini-STR (<220 bp) | 10 | ||
| Gender markers | Y-indel, amelogenin, and DYS391 | ||
| IQC markers | No | Yes: for distinguishing inhibited and degraded sample; positive control for PCR amplification | No |
| Identical primer sequences | ✓ | ✓ (except for the IQC) | ✓ |
| Probability of identity (PI) value | African American: 6.8 x 10-27 | ||
| US Caucasian: 3.71 x 10-26 | |||
| US Hispanic: 3.09 x 10-26 | |||
| Asian: 3.24 x 10-24 | |||
| Required ESS markers | ✓ | ✓ | ✓ |
| Required CODIS markers | ✓ | ✓ | ✓ |
| NDIS* approved | ✓ | ✓ | ✓ |
| DNAinput | 15 μL/1 ng target | Treated or untreated paper: 1.2 punch Swab: 3 μL (of 400 L) Applied Biosystem Prep-n-Go Buffer | |
| Final PCR volume | 25 μL | 15 μL | |
| Technical note supporting direct amplification | ✓ | No | ✓ |
| Supported sample types | Optimized chemistry for challenging samples types: touched, inhibited, or degraded samples | Verified with multiple sample collection devices such as treated paper, untreated paper, and swabs; designed to work with the most commonly used substrates | |
Featured video
Dr. Antonio Alonso discusses how forensic laboratories can make the transition to 6-dye chemistries in order to gain workflow efficiency and reduce operating costs.
Featured blog post and study
Recovering maximum information from degraded samples
In order to enhance information recovery from challenging bone samples, a recent degradation study was conducted by the University of North Texas Health Science Center, Department of Forensic and Investigative Genetics, US.
Featured documents
Need help bringing products online?
Look no further than our Human Identification Professional Services (HPS) team. Since 2007, HPS has performed over 2,400 services for over 350 customers across 55 countries. HPS’s wide array of service offerings includes validation, training, performance checks, and implementations based on internationally recognized standards. Technical project managers and field specialists bring extensive knowledge and experience to each project, helping seamlessly implement new workflows while minimizing impact on daily operations.
Resources & FAQs
- National Institute of Toxicology & Forensic Sciences, Madrid: Globalfiler Kit Validation
- Saint Louis County Police Crime Lab: GlobalFiler Kit Validation
- Saint Louis County Police Crime Lab: Interesting Cases
- Kansas City Police Crime Laboratory: Validation of GlobalFiler, Quantifiler on the 3500 GA
- HID Professional Services (HPS) tips and tricks for validations of the GlobalFiler™ Kit
Wu, L., et al (2017). Population genetic analysis of Xiamen Han population on 21 short tandem repeat loci
Customer support
If you have additional comments or questions, please contact the customer service team via email.
Technical support
If you are a current customer and need technical support, please contact the global service team via email, or phone us at 1-888-821-4HID (4443). Select option #1 for immediate assistance.
For Research Forensic or Paternity Use Only. Not for use in diagnostic procedures. For licensing and limited use restrictions visit thermofisher.com/HIDlicensing