Search
microRNAs: Processing
Several hundred miRNAs have been cloned and sequenced from mouse, human, Drosophila, C. elegans, and Arabidopsis (see www.sanger.ac.uk). Estimates suggest that 200–300 unique miRNA genes are present in the genomes of humans and mice (Lim 2003 b). The sequences of many of the miRNAs are homologous among organisms, suggesting that miRNAs represent a relatively old and important regulatory pathway (Grosshans 2002).
Most of the genome sequences encoding miRNAs occur in areas of the genome that are not associated with known genes; many are found in fragile sites in human chromosomes (Calin 2004) and appear to be independently transcribed (Lagos-Quintana 2001, Lau 2001, Lee 2001, Lim 2003a, 2003b). A number of miRNAs, are encoded in introns of primary mRNA transcripts. Typically they are encoded in the same orientation as the parent transcript, indicating that transcription of this class of miRNA gene is driven by an mRNA promoter (Aravin 2003, Lagos-Quintana 2003, Lai 2003, Lim 2003a). Unlike C. elegans and human, in Drosophila, most miRNA genes are found as clusters in the genome. There is good evidence that these clustered miRNA genes are expressed as multi-cistronic transcripts which are then processed to become mature miRNAs.
The excision and activation of active single-stranded miRNAs from precursor transcripts occurs through a multi-step process that is depicted in Figure 2, and is described below.
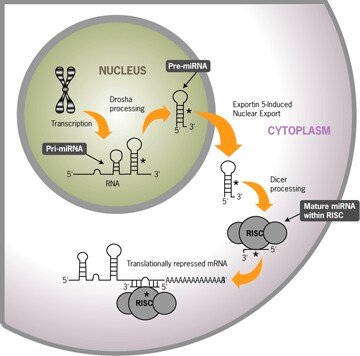
miRNA Processing and Activity
1. Transcription
miRNAs are initially expressed as part of transcripts termed primary miRNAs (pri-miRNAs) (Lee 2002). They are apparently transcribed by RNA Polymerase II, and include 5' caps and 3' poly(A) tails (Smalheiser 2003, Cai 2004). The miRNA portion of the pri-miRNA transcript likely forms a hairpin with signals for dsRNA-specific nuclease cleavage.
2. Hairpin release in the nucleus
The dsRNA-specific ribonuclease Drosha digests the pri-miRNA in the nucleus to release hairpin, precursor miRNA (pre-miRNA) (Lee 2003). Pre-miRNAs appear to be approximately 70 nt RNAs with 1–4 nt 3' overhangs, 25–30 bp stems, and relatively small loops. Drosha also generates either the 5' or 3' end of the mature miRNA, depending on which strand of the pre-miRNA is selected by RISC (Lee 2003, Yi 2003).
3. Export to the cytoplasm
Exportin-5 (Exp5) seems to be responsible for export of pre-miRNAs from the nucleus to the cytoplasm. Exp5 has been shown to bind directly and specifically to correctly processed pre-miRNAs. It is required for miRNA biogenesis, with a probable role in coordination of nuclear and cytoplasmic processing steps. (Lund 2003, Yi 2003).
4. Dicer processing
Dicer is a member of the RNase III superfamily of bidentate nucleases that has been implicated in RNA interference in nematodes, insects, and plants. Once in the cytoplasm, Dicer cleaves the pre-miRNA approximately 19 bp from the Drosha cut site (Lee 2003, Yi 2003). The resulting double-stranded RNA has 1–4 nt 3' overhangs at either end (Lund 2003). Only one of the two strands is the mature miRNA; some mature miRNAs derive from the leading strand of the pri-miRNA transcript, and with other miRNAs the lagging strand is the mature miRNA.
5. Strand selection by RISC
To control the translation of target mRNAs, the double-stranded RNA produced by Dicer must strand separate, and the single-stranded mature miRNA must associate with the RISC (Hutvagner 2002). Selection of the active strand from the dsRNA appears to be based primarily on the stability of the termini of the two ends of the dsRNA (Schwarz 2003, Khvorova 2003). The strand with lower stability base pairing of the 2–4 nt at the 5' end of the duplex preferentially associates with RISC and thus becomes the active miRNA (Schwarz 2003).
References
Aravin AA, Lagos-Quintana M, Yalcin A, Zavolan M, Marks D, Snyder B, Gaasterland T, Meyer J, and Tuschl T (2003) The small RNA profile during Drosophila melanogaster development. Dev Cell 5: 337–350.
Cai X, Hagedorn CH, Cullen BR. (2004) Human microRNAs are processed from capped, polyadenylated transcripts that can also function as mRNAs. RNA. Nov 3 [Epub ahead of print]
Calin GA, Sevignani C, Dumitru CD, Hyslop T, Noch E, Yendamuri S, Shimizu M, Rattan S, Bullrich F, Negrini M, Croce CM (2004) Human miRNA genes are frequently located at fragile sites and genomic regions involved in cancers. Proc. Natl. Acad. Sci. USA. 101: 2999-3004.
Grosshans H & Slack F (2002) Micro-RNAs: Small is plentiful. J Cell Biol 156: 17-21.
Hutvagner G, Zamore PD (2002) A miRNA in a multiple-turnover RNAi enzyme complex. Science 297, 2056-2060.
Khvorova A, Reynolds A, Jayasena SD. (2003) Functional siRNAs and miRNAs exhibit strand bias. Cell 115(2): 209-16.
Lai EC, Tomancak P, Williams RW, Rubin GM. (2003) Computational identification of Drosophila microRNA genes. Genome Biol 4(7): R42. Epub
Lagos-Quintana M, Rauhut R, Meyer J, Borkhardt A, Tuschl T (2003) New microRNAs from mouse and human. RNA 9: 175-179.
Lagos-Quintana M, Rauhut R, Lendeckel W, Tuschl T (2001) Identification of novel genes coding for small expressed RNAs. Science 294: 853-858.
Lau NC, Lim LP, Weinstein EG, Bartel DP (2001) Science 294: 858-62.
Lee, R. C. and Ambros, V. (2001) An extensive class of small RNAs in Caenorhabditis elegans. Science 294: 862-864.
Lee Y, Jeon K, Lee JT, Kim S, Kim VN (2002) MiRNA maturation: stepwise processing and subcellular localization. EMBO J 21: 4663-4670.
Lee Y, Ahn C, Han J, Choi H, Kim J, Yim J, Lee J, Provost P, Radmark O, Kim S, Kim VN (2003) The nuclear RNase III Drosha initiates miRNA processing. Nature 425: 415-419.
Lim LP, Lau NC, Weinstein EG, Abdelhakim A, Yekta S, Rhoades MW, Burge CB, Bartel DP (2003a) The microRNAs of Caenorhabditis elegans. Genes and Development 17: 991-1008.
Lim LP, Glasner ME, Yekta S, Burge CB, Bartel DP (2003b) Vertebrate microRNA genes. Science 299: 1540.
Lund E, Guttinger S, Calado A, Dahlberg JE, Kutay U. (2003) Nuclear Export of MicroRNA Precursors. Science. 2003 Nov 20 [Epub ahead of print].
Smalheiser NR. (2003) EST analyses predict the existence of a population of chimeric microRNA precursor-mRNA transcripts expressed in normal human and mouse tissues. Genome Biol 4(7): 403. Epub
Schwarz DS, Hutvagner G, Du T, Xu Z, Aronin N, Zamore PD. (2003) Asymmetry in the assembly of the RNAi enzyme complex. Cell 115(2): 199-208.
Yi R, Qin Y, Macara IG, Cullen BR (2003) Exportin-5 mediates the nuclear export of pre-microRNAs and short hairpin RNAs. Genes Dev 17: 3011-3016.