Search
Tools for Blot Hybridization
Maximizing Blot Sensitivity
A primary limitation of all blot hybridizations is the efficiency of hybridization between the nucleic acids on the membrane, and the labeled nucleic acids in the hybridization solution. Research at Ambion indicates that under typical blot hybridization conditions, only 0.5-5% of the target molecules on a membrane are actually bound by available probe. This is consistent with published work [1]. This limitation reduces the sensitivities of blot-associated assays by 20–100-fold, greatly reducing the ability to detect rare messages with Northern blots and dot blots, or to analyze limited DNA samples by Southern blotting. In many cases, maximizing the sensitivity of blot hybridization would forestall the need to switch to PCR-based detection methods, which are more difficult to set up and potentially less quantitative.
Ambion has developed a hybridization buffer that maximizes hybridization efficiency without increasing non-specific background. Our ultrasensitive hybridization buffer, ULTRAhyb, is as much as 100 times more sensitive than commonly used hybridization buffers (Figure 1). The benefits of greater sensitivity are remarkable. Using ULTRAhyb with Northerns, mRNAs too rare to detect using standard means are detected in an overnight exposure. Single copy genes are readily detected in only 1 µg of genomic DNA by Southerns only compared to the 10 µg typically required when using standard hybridization buffers.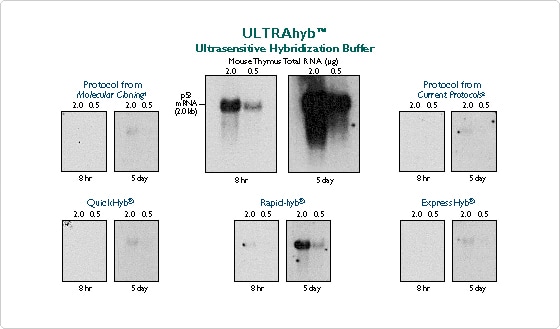
Figure 1. ULTRAhyb Detects a Signal in Only 8 Hours, Compared to a Minimum of 5 Days Using Competitors' Hybridization Solutions. Six identical Northern blots were assayed for p53 using 106 cpm/ml of a random-primed, labeled probe. The blots were incubated in the hybridization buffers indicated, following the manufacturer's recommendations for time and temperature. All blots were washed identically using 2X SSC/0.1% SDS and 0.1X SSC/0.1% SDS. The blots were exposed to the same piece of film at -80°C with one intensifying screen for the indicated times.
1. Molecular Cloning: A Laboratory Manual, 1989. Sambrook, J. Fritsch, E.F., Maniatis, T. Cold Spring Harbor Laboratory Press.
2. Current Protocols in Molecular Biology, 1994. Ausubel, F.M. and others, editors. John Wiley & Sons, Inc.
QuikHyb, Rapid-hyb and ExpressHyb are registered trademarks of Stratagene, Amersham and CLONTECH, respectively.
Reagents for Northern and Southern Blotting
Although Ambion's positively charged nylon membranes and wash buffers were developed primarily for Northern blot applications, they are also well-suited for other types of blots (e.g., Southerns, dot/slot blots, gene arrays).
Transfer
NorthernMax Rapid Transfer Buffer offers maximum transfer of RNA from agarose gels to membranes in two to three hours, regardless of size. Ambion's NorthernMax procedure uses downward passive transfer. Downward transfer does not crush the gel and makes use of gravity to speed the transfer process. The alkaline NorthernMax Rapid Transfer Buffer nicks the mRNA in the gel, reducing even the longest transcripts to molecules that can be effectively transferred to membrane, ensuring that all RNAs transfer with equal efficiency.
Membranes
Ambion's positively charged nylon membrane, BrightStar-Plus, has the lowest background and highest target retention of all membranes that we have tested. Each lot of membrane is compared to previous lots for consistency by our Quality Control Department, a claim that other molecular biology companies can not make.
RNase-free Wash Buffers
Ambion's Low and High Stringency Wash buffers are optimized to reduce background and cross-hybridization without decreasing hybridization signal. Each lot of blot hybridization products is functionally tested by our Quality Control Department to assure the highest possible sensitivity and lowest background. In addition, all solutions are certified nuclease-free.
Millennium Markers
Northern blots are used extensively to assess mRNA size. Ambion offers Millennium Markers, a set of evenly spaced, single-stranded RNA transcripts to help accurately determine mRNA size. The Millennium Marker mixture includes 0.5, 1, 1.5, 2, 2.5, 3, 4, 5, 6, and 9 kilobase transcripts that can be radioactively labeled, probed with our Millennium Marker Probe, or ethidium bromide stained to provide an accurate sizing ladder in gels or on autoradiographs.
Compatible Reagents for Blot Hybridization
References
For Research Use Only. Not for use in diagnostic procedures.