Search
siRNA-Mediated Gene Silencing in Mammalian Cells
Highlights
- Over half of the siRNAs tested provide greater than 50% reduction in target gene product
- Positive correlation exists between mRNA and protein suppression in genes targeted by siRNAs
- Enzymatically prepared siRNAs proved to be more potent than chemically synthesized siRNAs
- siRNA-induced gene silencing occurs in diverse mammalian cell types
Introduction
Multiple small interfering RNAs (siRNAs) complementary to c-myc and GAPDH mRNA were evaluated to establish parameters for siRNA design, to optimize transfection procedures for short, double-stranded RNAs, and to develop a new method for siRNA synthesis. Using a novel method of siRNA synthesis, four siRNAs were prepared that spanned the length of each target gene. These siRNAs were transfected into several types of mammalian cells and the relative reduction in target RNA and protein was determined. siRNAs to different target sites provided different levels of mRNA and protein knockdown. Spatial orientation along the length of the mRNA appeared not to be a factor in dictating siRNA activity, but target sites with lower GC content tended to be more susceptible. Less in vitro transcribed siRNA than chemically synthesized siRNA was needed for transfection. The most effective GAPDH siRNA was used to develop transfection protocols for various cell types. The novel method of siRNA synthesis used here provides a cost effective alternative to chemical synthesis.
Experimental Method
- siRNAs to human GAPDH, c-myc, and La were designed following the procedure described by Elbashir et al. (2001).
- siRNAs were prepared either by in vitro transcription using the Ambion Silencer™ siRNA Construction Kit or by chemical synthesis.
- After the addition of siRNAs and transfection reagent, cells were incubated 8-72 hr before harvesting and analysis.
- Target mRNA expression levels were determined by either Northern analysis using the NorthernMax-Gly Kit (Ambion) or by real-time RT-PCR on the ABI7700 with dual labeled probe detection.
- Protein levels were measured in experimental samples by Western analysis or by immunofluorescence analysis (antibody details).
- Cell proliferation assays were performed using Alamar Blue (Biosource International, Inc.)
Results
| siRNA Target Location Target | mRNA Expression Target | Protein Expression | |
|---|---|---|---|
| c-myc |
|
|
|
| GAPDH |
|
|
|
| La |
|
|
|
Table 1. Reduction in Target mRNA and Protein Induced by siRNAs.
siRNAs to different sequences within a gene are not equally effective at reducing gene expression. (Figures 1 and 2). 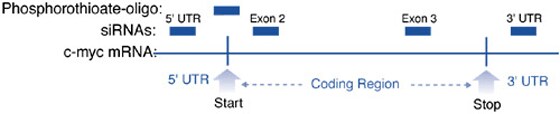
Figure 1. Locations of the siRNAs in c-myc mRNA. The c-myc mRNA is depicted as a line that is divided into 5' UTR, coding region, and 3' UTR. Also shown are the start and stop codons and the location of the phosphorothioate oligonucleotide. 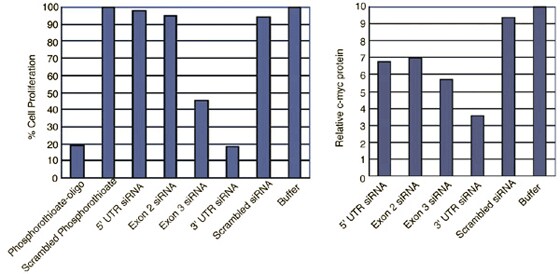
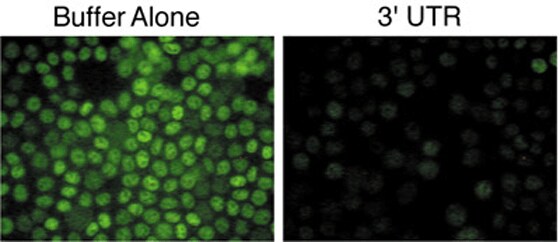
Figure 2. siRNAs have Differential Abilities to Induce RNAi. (A) siRNAs designed to different regions of the c-myc RNA sequence were transfected into the HeLa S3 cells and analyzed 48 hr after transfection for changes in proliferation and protein levels. Cell proliferation rates were analyzed using the Alamar Blue assay. (B) Immunofluorescence analysis of c-myc protein in HeLa S3 cells 48 hr following transfection with buffer only or with the siRNA against the 3' UTR.
The extent of gene silencing is apparent at both mRNA and protein levels. (Figures 2, 3 and 4). 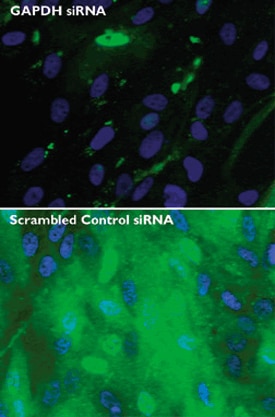
![]()
Figure 3. Reduction of GAPDH protein by in vitro transcribed siRNA. Immunhistochemistry staining of BJ fibroblast cells with fluorescein-labeled monoclonal antibody to GAPDH and DAPI nuclei staining 48 hr following transfection with scrambled control siRNA or siRNA against GAPDH. 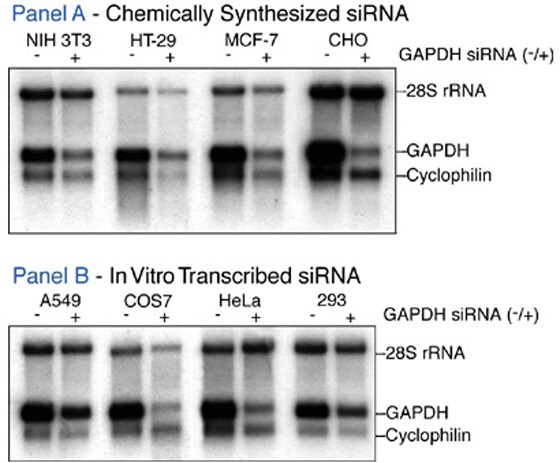
Figure 4. Susceptibility of Various Mammalian Cells to siRNA-induced Gene Silencing. The indicated cell types were transfected with a chemically synthesized (Panel A) or in vitro transcribed (Panel B) siRNA specific to GAPDH. The cells were harvested 48 hr after transfection. RNA was isolated, fractionated by denaturing gel electrophoresis, and transferred to a nylon membrane. The Northern blots were hybridized with RNA probes specific to GAPDH, cyclophilin, and 28S rRNA. The 28S rRNA probe was made to significantly lower specific activity.
siRNAs suppressed intended targets with no apparent nonspecific effects on unrelated genes. (Figure 4).
All cell types tested were susceptible to some level of silencing by the GAPDH siRNA. (Figure 4).
The optimal concentration for transfection of in vitro transcribed siRNA was consistently at least 10-fold lower than that for chemically synthesized RNA. (Figure 5). 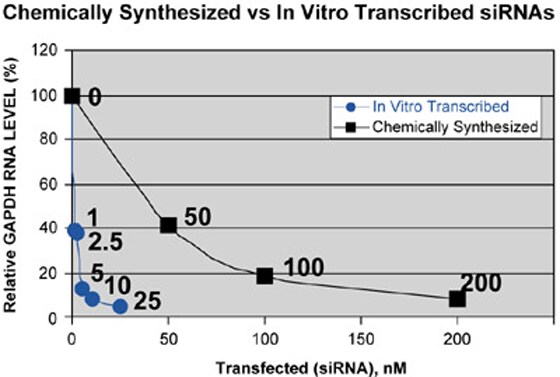
Figure 5. Potency of siRNAs Prepared by Chemical or Enzymatic Synthesis. HeLa cells were transfected with the indicated concentration of GAPDH siRNA prepared by chemical synthesis (synthetic) or in vitro transcription. Both siRNAs were made to the same sequence of the mRNA. GAPDH mRNA levels from the various samples were assessed by Northern analysis. GAPDH signals were qualified using a phosphorimager and normalized using signal from a cyclophilin probe.
Discussion
Antibodies
- The mouse monoclonal antibody to GAPDH was from RDI (Cat #RDI-TRK5G4-6C5).
- The mouse monoclonal antibody to La was from BD Transduction Laboratories (Cat #610904).
- The mouse monoclonal antibody for c-myc was from NeoMarkers (c-myc Ab-5 clone 67P05).
Reference
- Elbashir SM, Harborth J, Lendeckel W, Yalcin A, Weber K, Tuschl T (2001) Nature 411: 494-498.