Search
Gene silencing by RNA interference is being used routinely to study gene function in cultured mammalian cells
While this approach has been extremely powerful, it does not allow a critical evaluation of how genes function within the whole organism. To address this problem, researchers are now applying RNAi in vivo. Although still in its early stages, use of RNAi in vivo already shows promise and significant advances have been made.
The two basic methods
Two basic methods for triggering RNAi have been adapted for use in vivo: delivery of siRNAs, and delivery of plasmid and viral vectors that express a short hairpin RNA (shRNA) that is subsequently processed into active siRNA. As with the application of RNAi in cultured cells, use of siRNAs is more prominent than use of shRNA expression vectors. For RNAi experiments in cultured cells, effective siRNAs to a particular target of interest are easier to obtain, and because they are small and only need to cross the cell membrane and not the nuclear membrane to be effective, they are easier to deliver. In contrast, shRNA expression vectors are time consuming to construct, particularly when one takes into account the time required to create and test several shRNA sequences to find an effective one. siRNAs are also easier to deliver than plasmid based shRNA expression vectors, and they do not have the associated problems with insertional mutagenesis and immunogenicity that plague retroviral (including lentiviral) and adenoviral vectors, respectively.
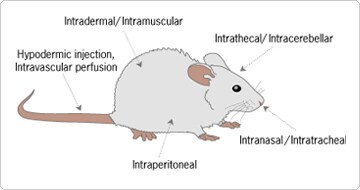
Figure 1. Strategies for Delivery of siRNA Molecules In Vivo.
The success of siRNA mediated gene silencing in vivo depends on efficient delivery and retention of the siRNA in the vasculature of a specific tissue of interest, and its effective uptake by those cells. In addition, the siRNA must remain stable until it can reach its ultimate destination. Although early, there have been reports of success using both local and systemic delivery of siRNAs.
Success with local administration of siRNAs
Perhaps the greatest success has come with local administration of siRNA to the eye. An siRNA to VEGF delivered to the subretinal space in mice has been proven to reduce eye angiogenesis [1]. In this model, there was no need to modify the siRNA, or to complex it with lipid polymer or other delivery agents, as has been necessary in some other systems. More recently, an siRNA to VEGF, developed by Acuity Pharmaceuticals and designed to be delivered via intravitreal injection, has entered phase II clinical trials as a treatment for wet age-related macular degeneration.
In rat models, modified siRNAs to the pain related cation channel P2X3 have been successfully delivered into the brains of rats via intrathecal infusion using surgically implanted pumps; the siRNAs significantly inhibited the neuropathic pain response in this model system [2]. Although not practical for many researchers, this approach demonstrates that siRNAs can be successfully delivered locally into the rodent brain, and that in vivo functional genomics studies of CNS related genes using siRNAs are possible.
There is considerable interest in delivering siRNAs into the lung in the attempts to study pulmonary as well as infectious diseases and to potentially treat influenza, SARS, and other clinically relevant pulmonary diseases caused by RNA viruses. In one promising study, an intranasal delivery system was used in primates to deliver a SARS virus specific siRNA, resulting in reduced fever, decreased viral load, and reduced alveoli damage [3]. This study demonstrates the validity of intranasal delivery of siRNAs to the lung.
Progress with systemic siRNA delivery
The feasibility of systemic in vivo siRNA delivery in mammals was first demonstrated using hydrodynamic tail vein injections† in mice. In this procedure, unmodified siRNAs are rapidly injected into the tail vein in a large volume of aqueous solution, resulting in localization within hepatocytes [3]. Although not clinically relevant, this procedure does permit gene function and drug target validation studies within the rodent liver, until more effective delivery technologies are developed. In support of this technique, Song and colleagues found that hydrodynamic injection of a Fas siRNA resulted in silencing of Fas in mouse hepatocytes for a period of 10 days. This treatment protected hepatocytes from Fas antibody and concanavalin A stimulated apoptosis, and protected mice from fulminant hepatitis [4]. Similarly, hydrodynamic tail vein injection of a caspase 8 siRNA protected mice against acute liver failure induced by Fas antibody or expression of Fas ligand [5].
More recently, low volume, normal pressure intravenous delivery of a modified siRNA targeting apolipoprotein B in mice resulted in gene silencing in the liver and jejunum. The siRNA was conjugated with cholesterol to provide targeted delivery, and included backbone and sugar modifications to enhance serum stability [6].
Systemic delivery of siRNAs will likely be required to target most tumor types, as well as many other in vivo targets. To this end, several groups are investigating the use of lipid based and nanoparticle based siRNA delivery complexes [7-10].
The work on systemic delivery of siRNAs illustrates three obstacles that must be overcome for siRNA to be successful in vivo: selective delivery into the desired tissue, adequate protection from degradation en route to the target tissue, and protection of the siRNA from rapid excretion. Surprisingly, rapid excretion has proven to be more of a problem than in vivo stability [11]. Although chemical stabilization is readily achieved via siRNA modification, it does not appear to be necessary in most cases, as excretion appears to occur prior to degradation. Use of nanoparticles or lipid complexes currently shows more promise than chemical modification to address the pharmacokinetics and tissue distribution issues endemic to in vivo siRNA delivery.
Looking to the future
RNAi is now firmly entrenched as an invaluable tool in most drug discovery pipelines, and further advances in the technology will no doubt enhance the utility of the technique in identifying and validating potential drug targets in vivo. In addition, siRNAs themselves have huge potential as therapeutic agents. If realized, the impact on the pharmaceutical industry would be revolutionary. The most significant hurdle for the therapeutic use of siRNAs is how to provide targeted delivery. Although significant progress has been made, delivery of nucleic acids to specific organs, tissues, and cells will require additional advances, including development of possible novel conjugations and/or formulations to specifically target certain cells.
†In vivo hydrodynamic delivery of nucleic acids is covered by patents and patent applications of Mirus Bio Corporation, including U.S. Patents 6,627,616, 6,379,966 and 6,897,068 and related filings worldwide. Research and commercial uses by for-profit entities require a license--please see www.mirusbio.com for contact information.
Silencer In Vivo ready siRNAs
To support the growing use of siRNAs in vivo, Ambion now provides a new grade of siRNAs called Silencer In Vivo Ready siRNAs that are specifically for research use in animals. These siRNAs feature:
- Convenient 100, 250, and 1000 nmol sizes; larger sizes available upon request
- HPLC purified to >95% purity
- Free of harmful salts
- Endotoxin tested
- Reich SJ, Fosnot J, Kuroki A, Tang W, Yang X, Maguire AM, Bennett J, Tolentino MJ (2003) Small interfering RNA (siRNA) targeting VEGF effectively inhibits ocular neovascularization in a mouse model. Mol Vis 9:210–6.
- Dorn G, Patel S, Wotherspoon G, Hemmings-Mieszczak M, Barclay J, Natt FJ, Martin P, Bevan S, Fox A, Ganju P, Wishart W, Hall J (2004) siRNA relieves chronic neuropathic pain. Nucleic Acids Res 32(5):e49.
- Li BJ, Tang Q, Cheng D, Qin C, Xie FY, Wei Q, Xu J, Liu Y, Zheng BJ, Woodle MC, Zhong N, Lu PY (2005) Using siRNA in prophylactic and therapeutic regimens against SARS coronavirus in Rhesus macaque. Nat Med 11(9):944–51.
- Song E, Lee SK, Wang J, Ince N, Ouyang N, Min J, Chen J, Shankar P, Lieberman J (2003) RNA interference targeting Fas protects mice from fulminant hepatitis. Nat Med 9(3):347–51.
- Zender L, Hutker S, Liedtke C, Tillmann HL, Zender S, Mundt B, Waltemathe M, Gosling T, Flemming P, Malek NP, Trautwein C, Manns MP, Kuhnel F, Kubicka S (2003) Caspase 8 small interfering RNA prevents acute liver failure in mice. Proc Natl Acad Sci USA 100(13):7797–802.
- Soutschek J, Akinc A, Bramlage B, Charisse K, Constien R, Donoghue M, Elbashir S, Geick A, Hadwiger P, Harborth J, John M, Kesavan V, Lavine G, Pandey RK, Racie T, Rajeev KG, Rohl I, Toudjarska I, Wang G, Wuschko S, Bumcrot D, Koteliansky V, Limmer S, Manoharan M, Vornlocher HP (2004) Therapeutic silencing of an endogenous gene by systemic administration of modified siRNAs. Nature 432(7014):173–8.
- Urban-Klein B, Werth S, Abuharbeid S, Czubayko F, Aigner A (2005) RNAi-mediated gene-targeting through systemic application of polyethylenimine (PEI)-complexed siRNA in vivo. Gene Ther 12(5):461–6.
- Pal A, Ahmad A, Khan S, Sakabe I, Zhang C, Kasid UN, Ahmad I (2005) Systemic delivery of Raf siRNA using cationic cardiolipin liposomes silences Raf-1 expression and inhibits tumor growth in xenograft model of human prostate cancer. Int J Oncol 26(4):1087–91.
- Morrissey DV, Lockridge JA, Shaw L, Blanchard K, Jensen K, Breen W, Hartsough K, Machemer L, Radka S, Jadhav V, Vaish N, Zinnen S, Vargeese C, Bowman K, Shaffer CS, Jeffs LB, Judge A, MacLachlan I, Polisky B (2005) Potent and persistent in vivo anti-HBV activity of chemically modified siRNAs. Nat Biotechnol 23(8):1002–7.
- Schiffelers RM, Ansari A, Xu J, Zhou Q, Tang Q, Storm G, Molema G, Lu PY, Scaria PV, Woodle MC (2004) Cancer siRNA therapy by tumor selective delivery with ligand-targeted sterically stabilized nanoparticle. Nucleic Acids Res 32(19):e149.
- Lu PY, Xie F, Woodle MC (2005) In vivo application of RNA interference: from functional genomics to therapeutics. Adv Genet 54:117–42.
RNAi resources
For Research Use Only. Not for use in diagnostic procedures.